Strontium, a silvery-white alkaline earth metal, holds a significant place in the periodic table. Its atomic number is 38, and it is represented by the symbol Sr. Strontium is known for its unique properties and various applications, making it a subject of interest for scientists, engineers, and enthusiasts alike. This blog post delves into the fascinating world of strontium, exploring its position on the periodic table, its chemical properties, and its diverse uses.
Strontium on the Periodic Table
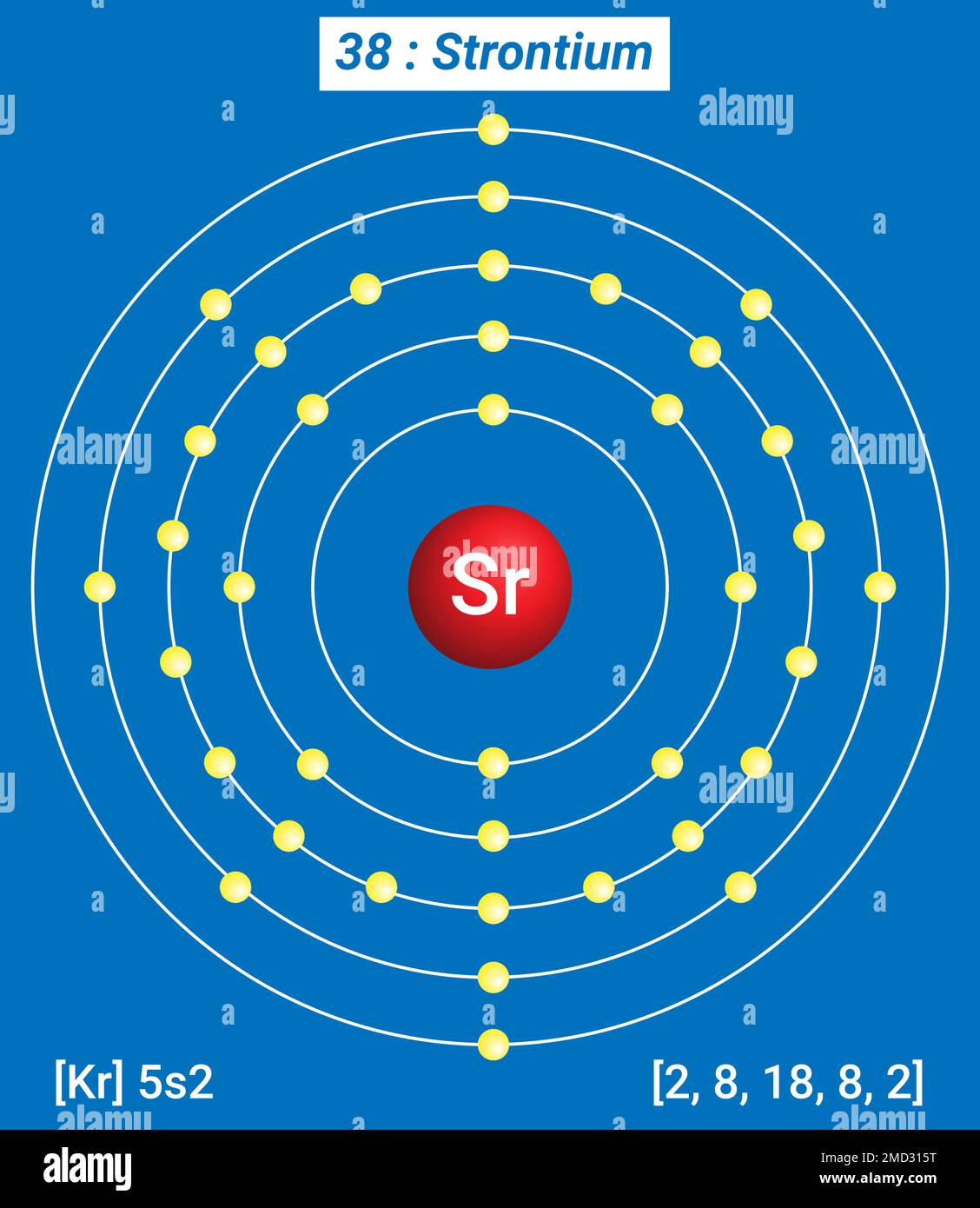
Strontium is located in Group 2 (alkaline earth metals) of the periodic table. It is positioned between calcium (Ca) and barium (Ba). This placement is crucial as it determines many of its chemical and physical properties. The periodic table is a systematic arrangement of elements based on their atomic numbers, electron configurations, and recurring chemical properties. Strontium's position in Group 2 means it has two electrons in its outermost shell, which it readily gives up to form positive ions.
Strontium's atomic number is 38, indicating it has 38 protons in its nucleus. Its electronic configuration is [Kr] 5s2, where [Kr] represents the electron configuration of krypton, a noble gas. This configuration explains why strontium behaves similarly to other alkaline earth metals, such as calcium and barium.
Chemical Properties of Strontium
Strontium exhibits several notable chemical properties that make it unique among the elements. Some of its key properties include:
- Atomic Weight: Approximately 87.62 u
- Melting Point: 777°C (1431°F)
- Boiling Point: 1384°C (2523°F)
- Density: 2.64 g/cm3 at room temperature
- Electronegativity: 0.95 on the Pauling scale
Strontium is highly reactive and can form compounds with various elements. It readily reacts with water to produce strontium hydroxide and hydrogen gas. This reactivity is a common trait among alkaline earth metals. Strontium also reacts with oxygen to form strontium oxide, which is a white powder used in various applications.
One of the most interesting properties of strontium is its ability to emit light when exposed to certain conditions. This property is utilized in fireworks and flares, where strontium compounds produce a bright red color. The emission of light is due to the excitation of electrons in the strontium atoms, which then release energy in the form of photons as they return to their ground state.
Applications of Strontium
Strontium's unique properties make it valuable in various industries. Some of the most significant applications of strontium include:
- Fireworks and Flares: Strontium compounds, such as strontium nitrate and strontium carbonate, are used to produce a vibrant red color in fireworks and flares. The bright red light emitted by strontium is highly visible, making it ideal for signaling and pyrotechnic displays.
- Glass Manufacturing: Strontium oxide is used in the production of certain types of glass, such as color television picture tubes and glass for color television screens. It helps to improve the clarity and durability of the glass.
- Ceramics: Strontium is used in the manufacture of ceramics, particularly in the production of ferrite magnets. These magnets are used in various electronic devices, including motors, generators, and magnetic recording media.
- Medical Applications: Strontium-89, a radioactive isotope of strontium, is used in medical treatments for bone cancer. It targets and destroys cancer cells in the bones, providing relief from pain and slowing the progression of the disease.
- Toothpaste: Strontium chloride is used in some toothpastes to help reduce tooth sensitivity. It works by blocking the tiny tubes in the dentin that lead to the nerve, reducing the sensation of pain.
Strontium's versatility extends beyond these applications. Its compounds are also used in the production of certain types of cement, in the refining of zinc, and in the manufacture of certain types of batteries. The diverse uses of strontium highlight its importance in modern industry and technology.
Environmental and Health Considerations
While strontium has many beneficial applications, it is also important to consider its environmental and health impacts. Strontium is naturally present in the environment, but exposure to high levels can have adverse effects. Some key considerations include:
- Environmental Impact: Strontium can enter the environment through natural processes and human activities, such as mining and industrial processes. High levels of strontium in soil and water can affect plant and animal life, although strontium is generally considered less toxic than other heavy metals.
- Health Effects: Exposure to strontium can occur through inhalation, ingestion, or skin contact. High levels of strontium can affect bone health, as strontium can replace calcium in the bones. This can lead to bone disorders and other health issues. However, the health effects of strontium are generally considered to be low compared to other heavy metals.
- Regulation and Safety: To minimize the risks associated with strontium, it is important to follow proper safety protocols and regulations. This includes using personal protective equipment, proper ventilation, and safe disposal methods for strontium-containing materials.
Strontium's environmental and health considerations are important to keep in mind, especially in industries where strontium is used extensively. By understanding the potential risks and taking appropriate precautions, the benefits of strontium can be maximized while minimizing its negative impacts.
Strontium in the Periodic Table
Strontium's position in the periodic table provides insights into its chemical behavior and properties. As an alkaline earth metal, strontium shares many characteristics with other elements in Group 2, such as calcium and barium. However, strontium also has unique properties that set it apart. For example, strontium's ability to emit light when excited makes it valuable in pyrotechnics and other applications.
Understanding strontium's place in the periodic table helps scientists and engineers predict its behavior in chemical reactions and develop new applications for this versatile element. The periodic table is a powerful tool for organizing and understanding the properties of the elements, and strontium is a prime example of how this organization can lead to practical and innovative uses.
Strontium's position in the periodic table also highlights the importance of the periodic table as a whole. The periodic table is not just a list of elements; it is a map of the chemical world, showing the relationships and patterns that govern the behavior of the elements. By studying strontium and its place in the periodic table, we gain a deeper understanding of the fundamental principles of chemistry and the natural world.
Strontium's unique properties and diverse applications make it a fascinating subject of study. From its use in fireworks and flares to its role in medical treatments and glass manufacturing, strontium plays a crucial role in modern industry and technology. By understanding strontium's position on the periodic table and its chemical properties, we can continue to explore new uses for this versatile element and unlock its full potential.
Strontium's position in the periodic table is a testament to the power of the periodic table as a tool for understanding the elements and their properties. By studying strontium and its place in the periodic table, we gain a deeper appreciation for the beauty and complexity of the chemical world. Whether you are a scientist, engineer, or enthusiast, strontium offers a wealth of opportunities for exploration and discovery.
📚 Note: The information provided in this blog post is for educational purposes only and should not be used as a substitute for professional advice. Always consult with a qualified expert before making decisions based on the information presented here.
Strontium’s journey from the periodic table to various applications is a testament to its versatility and importance. From its role in pyrotechnics to its use in medical treatments, strontium continues to play a crucial role in modern industry and technology. By understanding strontium’s properties and applications, we can appreciate the significance of this element and its place in the periodic table. As we continue to explore the chemical world, strontium will undoubtedly remain a subject of interest and discovery.
Related Terms:
- full electron configuration of strontium
- strontium periodic table symbol
- boiling point of strontium
- is strontium a metal
- 38th element periodic table
- strontium group