The Periodic Table With Spdf is a fundamental tool in chemistry that organizes elements based on their atomic number, electron configuration, and recurring chemical properties. The table is divided into periods (rows) and groups (columns), with each element's position reflecting its electronic structure. Understanding the Periodic Table With Spdf involves grasping the concept of electron shells and sub-shells, which are crucial for predicting an element's chemical behavior.
The Structure of the Periodic Table With Spdf
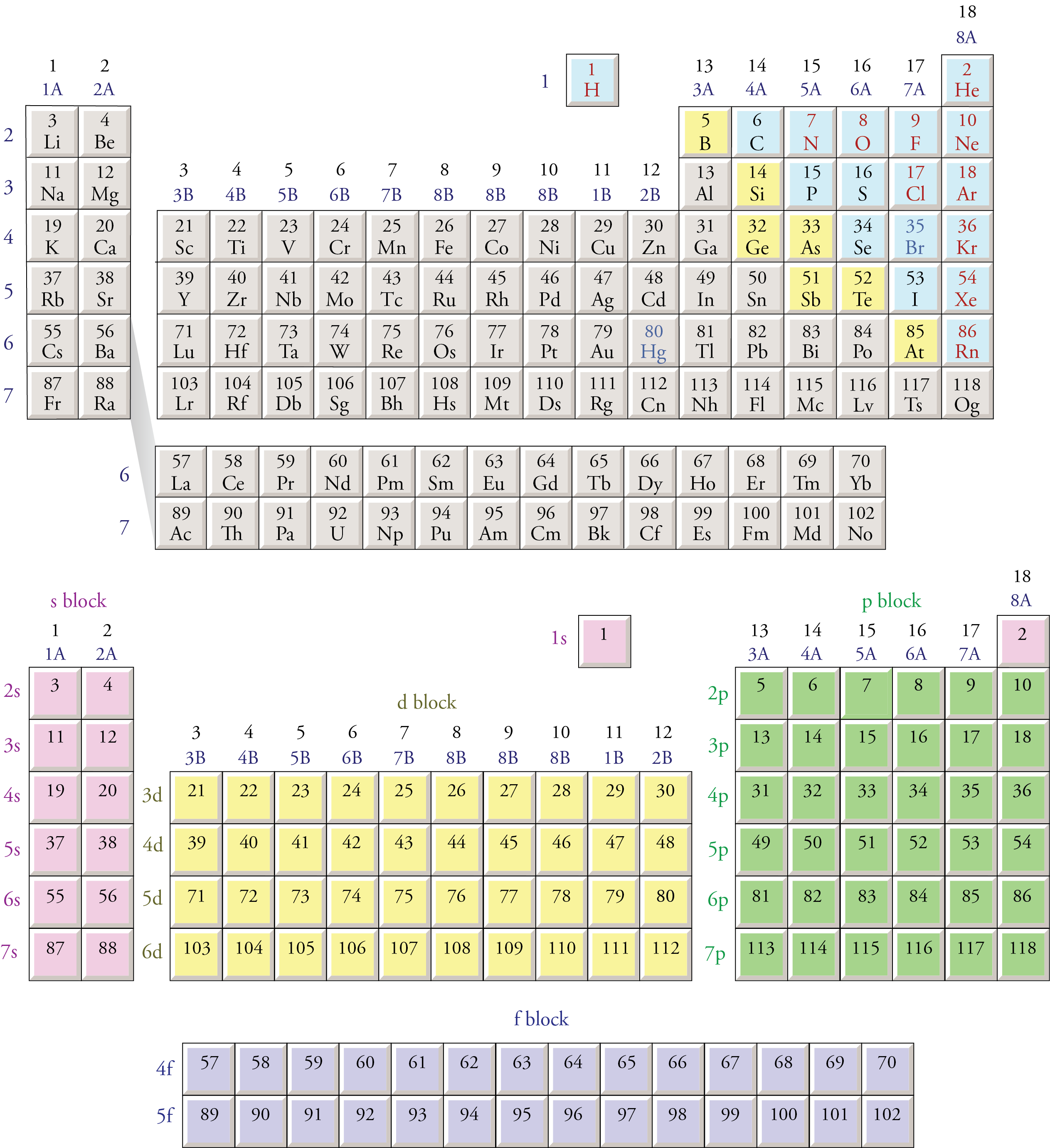
The Periodic Table With Spdf is structured to reflect the filling of electron shells and sub-shells. The table is divided into four main blocks: s-block, p-block, d-block, and f-block. Each block corresponds to the type of sub-shell being filled with electrons.
S-Block Elements
The s-block elements are found in groups 1 and 2 of the Periodic Table With Spdf. These elements have their outermost electrons in the s-sub-shell. Group 1 elements, known as alkali metals, have one electron in their outermost s-sub-shell, while group 2 elements, known as alkaline earth metals, have two electrons in their outermost s-sub-shell. These elements are highly reactive and tend to lose electrons easily.
P-Block Elements
The p-block elements are found in groups 13 to 18 of the Periodic Table With Spdf. These elements have their outermost electrons in the p-sub-shell. The p-block includes metals, metalloids, and nonmetals. The reactivity of p-block elements varies widely, from highly reactive metals like aluminum to inert gases like helium and neon.
D-Block Elements
The d-block elements, also known as transition metals, are found in groups 3 to 12 of the Periodic Table With Spdf. These elements have their outermost electrons in the d-sub-shell. Transition metals are known for their variable oxidation states and the ability to form colored compounds. They are often used in catalysis, electronics, and construction due to their unique properties.
F-Block Elements
The f-block elements, also known as lanthanides and actinides, are found at the bottom of the Periodic Table With Spdf. These elements have their outermost electrons in the f-sub-shell. Lanthanides and actinides are often referred to as inner transition metals. They have complex electronic structures and are known for their magnetic and electronic properties. Many of these elements are radioactive and are used in nuclear applications.
Electron Configuration and the Periodic Table With Spdf
Understanding the electron configuration of elements is crucial for interpreting the Periodic Table With Spdf. The electron configuration describes the arrangement of electrons in an atom's orbitals. The configuration is written using the notation nl, where n is the principal quantum number and l is the azimuthal quantum number.
The principal quantum number n indicates the energy level or shell, while the azimuthal quantum number l indicates the sub-shell. The sub-shells are designated by letters: s, p, d, and f. Each sub-shell can hold a specific number of electrons:
- s-sub-shell: 2 electrons
- p-sub-shell: 6 electrons
- d-sub-shell: 10 electrons
- f-sub-shell: 14 electrons
For example, the electron configuration of sodium (Na) is 1s22s22p63s1. This means that sodium has two electrons in the 1s sub-shell, two electrons in the 2s sub-shell, six electrons in the 2p sub-shell, and one electron in the 3s sub-shell.
Periodic Trends in the Periodic Table With Spdf
The Periodic Table With Spdf exhibits several periodic trends that help predict the properties of elements. These trends include atomic radius, ionization energy, electron affinity, and electronegativity.
Atomic Radius
The atomic radius is the distance from the nucleus to the outermost electron shell. As you move from left to right across a period, the atomic radius decreases due to the increasing nuclear charge, which pulls the electrons closer to the nucleus. As you move down a group, the atomic radius increases due to the addition of new electron shells.
Ionization Energy
Ionization energy is the energy required to remove an electron from an atom. As you move from left to right across a period, the ionization energy increases due to the increasing nuclear charge, which makes it harder to remove an electron. As you move down a group, the ionization energy decreases due to the increased distance between the outermost electrons and the nucleus.
Electron Affinity
Electron affinity is the energy released when an electron is added to a neutral atom. As you move from left to right across a period, the electron affinity generally increases due to the increasing nuclear charge, which attracts the incoming electron more strongly. As you move down a group, the electron affinity decreases due to the increased distance between the outermost electrons and the nucleus.
Electronegativity
Electronegativity is the ability of an atom to attract electrons in a chemical bond. As you move from left to right across a period, the electronegativity increases due to the increasing nuclear charge, which attracts electrons more strongly. As you move down a group, the electronegativity decreases due to the increased distance between the outermost electrons and the nucleus.
Applications of the Periodic Table With Spdf
The Periodic Table With Spdf is a powerful tool with numerous applications in chemistry, materials science, and other fields. Some of the key applications include:
- Predicting Chemical Properties: The table helps predict the chemical properties of elements based on their position and electron configuration.
- Designing New Materials: Understanding the periodic trends allows scientists to design new materials with specific properties.
- Catalysis: Transition metals, which are d-block elements, are widely used as catalysts in industrial processes.
- Nuclear Applications: Lanthanides and actinides, which are f-block elements, are used in nuclear reactors and weapons.
- Electronics: Elements like silicon and germanium, which are p-block elements, are used in semiconductors and electronic devices.
Examples of Elements in the Periodic Table With Spdf
Let's look at a few examples of elements from different blocks of the Periodic Table With Spdf to understand their properties better.
Sodium (Na)
Sodium is an s-block element with the electron configuration 1s22s22p63s1. It is a highly reactive alkali metal that readily loses its single 3s electron to form a positive ion (Na+). Sodium is used in various applications, including as a coolant in nuclear reactors and in the production of sodium vapor lamps.
Chlorine (Cl)
Chlorine is a p-block element with the electron configuration 1s22s22p63s23p5. It is a highly reactive nonmetal that readily gains an electron to form a negative ion (Cl-). Chlorine is used in water treatment, disinfection, and the production of various chemicals, including polyvinyl chloride (PVC).
Iron (Fe)
Iron is a d-block element with the electron configuration 1s22s22p63s23p64s23d6. It is a transition metal known for its strength, durability, and magnetic properties. Iron is widely used in construction, machinery, and as a catalyst in the Haber-Bosch process for ammonia production.
Uranium (U)
Uranium is an f-block element with the electron configuration 1s22s22p63s23p64s23d104p65s24f145d106s25f36d17s2. It is a radioactive actinide used as fuel in nuclear reactors and in nuclear weapons. Uranium's complex electronic structure makes it suitable for these applications.
📝 Note: The electron configurations provided are simplified and do not include the detailed filling of sub-shells according to the Aufbau principle and Hund's rule.
The Importance of the Periodic Table With Spdf in Education
The Periodic Table With Spdf is a fundamental tool in chemistry education. It helps students understand the organization of elements, their properties, and the trends that govern their behavior. By studying the Periodic Table With Spdf, students can:
- Learn about the structure of atoms and the arrangement of electrons in orbitals.
- Understand the periodic trends in atomic radius, ionization energy, electron affinity, and electronegativity.
- Predict the chemical properties of elements based on their position in the table.
- Appreciate the historical development of the periodic table and the contributions of scientists like Dmitri Mendeleev.
The Periodic Table With Spdf is not just a tool for chemists; it is a universal language that helps scientists and students communicate and understand the behavior of elements.

Periodic Table With Spdf and Modern Research
The Periodic Table With Spdf continues to be a vital tool in modern research. Scientists use the table to explore new elements, understand their properties, and develop new materials. Some of the recent advancements in the field include:
- Discovery of New Elements: The periodic table has been expanded to include elements with atomic numbers up to 118. These superheavy elements are synthesized in laboratories and have unique properties due to relativistic effects.
- Materials Science: Researchers use the periodic table to design new materials with specific properties, such as high-temperature superconductors, nanomaterials, and energy storage devices.
- Catalysis: Transition metals are extensively studied for their catalytic properties, leading to the development of more efficient and sustainable chemical processes.
- Nuclear Chemistry: The study of actinides and other radioactive elements is crucial for understanding nuclear reactions, developing nuclear fuels, and managing nuclear waste.
The Periodic Table With Spdf provides a framework for organizing and understanding the vast amount of data generated in these research areas. It helps scientists identify patterns, make predictions, and develop new theories.
Challenges and Future Directions
While the Periodic Table With Spdf is a powerful tool, it also faces challenges and limitations. Some of the key challenges include:
- Complexity of Superheavy Elements: The properties of superheavy elements are difficult to predict due to relativistic effects and the short half-lives of these elements.
- Incomplete Understanding of Electronic Structure: The electronic structure of some elements, particularly those with complex configurations, is not fully understood.
- Environmental and Safety Concerns: The use of certain elements, such as radioactive actinides, poses environmental and safety challenges.
Future research in the field of the Periodic Table With Spdf will focus on addressing these challenges and expanding our understanding of the elements. Some of the future directions include:
- Development of New Theoretical Models: Researchers are working on developing new theoretical models to better predict the properties of superheavy elements and other complex systems.
- Advanced Experimental Techniques: New experimental techniques, such as high-resolution spectroscopy and advanced computational methods, are being developed to study the electronic structure of elements.
- Sustainable Use of Elements: There is a growing emphasis on the sustainable use of elements, including the development of recycling technologies and the search for alternative materials.
The Periodic Table With Spdf will continue to evolve as our understanding of the elements deepens. It remains a cornerstone of chemistry and a testament to the power of human curiosity and ingenuity.
In conclusion, the Periodic Table With Spdf is an essential tool for understanding the properties and behavior of elements. It provides a framework for organizing elements based on their atomic number, electron configuration, and recurring chemical properties. By studying the Periodic Table With Spdf, we can predict the chemical properties of elements, design new materials, and explore the frontiers of modern research. The table’s structure, based on the filling of electron shells and sub-shells, helps us understand the periodic trends that govern the behavior of elements. Whether in education, research, or industry, the Periodic Table With Spdf remains a vital tool for scientists and students alike.
Related Terms:
- periodic table with spdf notation
- periodic table with spdf blocks
- printable electron configuration periodic table
- periodic table spdf labeled
- periodic table of elements spdf
- periodic table showing spdf