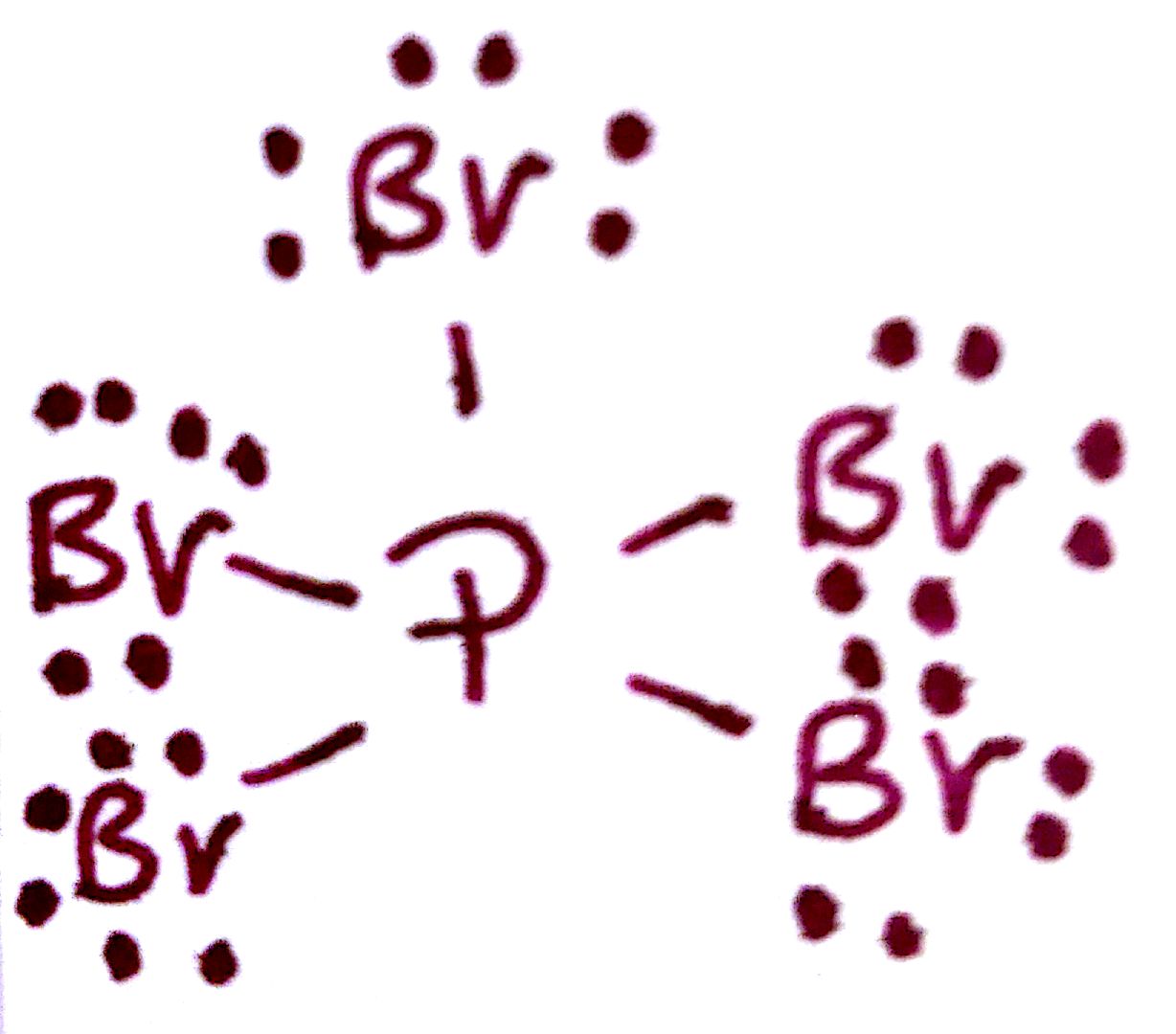
Understanding the Pbr5 Lewis Structure is crucial for anyone delving into the world of chemistry, particularly in the study of molecular geometry and bonding. The Lewis structure, also known as the Lewis dot diagram, is a simple way to represent the valence electrons in a molecule. It helps chemists visualize the bonding between atoms and predict the molecular shape. In this post, we will explore the Pbr5 Lewis Structure, its significance, and how to draw it step-by-step.
What is a Lewis Structure?
A Lewis structure is a diagram that shows the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. It was introduced by Gilbert N. Lewis in 1916 as a way to illustrate the arrangement of electrons in a molecule. The structure helps in understanding the chemical bonding and the molecular geometry.
The Importance of the Pbr5 Lewis Structure
The Pbr5 Lewis Structure is particularly important because it provides insights into the bonding and geometry of the molecule. Phosphorus pentabromide (PBr5) is a molecular compound that consists of one phosphorus atom and five bromine atoms. Understanding its Lewis structure helps in predicting its molecular shape, which is crucial for understanding its chemical properties and reactivity.
Steps to Draw the Pbr5 Lewis Structure
Drawing the Pbr5 Lewis Structure involves several steps. Follow these instructions to create an accurate Lewis structure for PBr5:
- Identify the central atom: In PBr5, the central atom is phosphorus (P).
- Count the total number of valence electrons: Phosphorus has 5 valence electrons, and each bromine atom has 7 valence electrons. Therefore, the total number of valence electrons is 5 (from P) + 5 * 7 (from Br) = 40.
- Draw the skeletal structure: Place the phosphorus atom in the center and the five bromine atoms around it.
- Form single bonds: Connect each bromine atom to the central phosphorus atom with a single bond. Each single bond consists of 2 electrons, so 5 * 2 = 10 electrons are used for bonding.
- Distribute the remaining electrons: After forming the bonds, 40 - 10 = 30 electrons are left. These electrons are distributed as lone pairs around the bromine atoms. Each bromine atom will have 3 lone pairs (6 electrons), and the phosphorus atom will have no lone pairs.
📝 Note: The phosphorus atom in PBr5 does not have any lone pairs because all its valence electrons are used in bonding.
Understanding the Pbr5 Lewis Structure
Once you have drawn the Pbr5 Lewis Structure, it is essential to understand its implications. The structure shows that phosphorus is surrounded by five bromine atoms, forming a trigonal bipyramidal geometry. This geometry is crucial for understanding the molecular properties and reactivity of PBr5.
Molecular Geometry of Pbr5
The molecular geometry of PBr5 is trigonal bipyramidal. In this geometry, the phosphorus atom is at the center, and the five bromine atoms are positioned at the vertices of a trigonal bipyramid. This arrangement minimizes electron repulsion and provides stability to the molecule.
Here is a table summarizing the key points of the PBr5 Lewis structure and its molecular geometry:
| Aspect | Description |
|---|---|
| Central Atom | Phosphorus (P) |
| Total Valence Electrons | 40 |
| Bonding Electrons | 10 (5 single bonds) |
| Lone Pairs on Bromine | 3 lone pairs per bromine atom |
| Molecular Geometry | Trigonal bipyramidal |
Applications of the Pbr5 Lewis Structure
The Pbr5 Lewis Structure has several applications in chemistry. It helps in understanding the reactivity of PBr5, predicting its chemical properties, and designing experiments. For example, knowing the molecular geometry of PBr5 can help in predicting its reactivity with other molecules and designing catalysts for chemical reactions.
Common Misconceptions About the Pbr5 Lewis Structure
There are several misconceptions about the Pbr5 Lewis Structure that need to be addressed. One common misconception is that the phosphorus atom in PBr5 has lone pairs of electrons. However, as mentioned earlier, all the valence electrons of phosphorus are used in bonding, and it does not have any lone pairs.
Another misconception is that the molecular geometry of PBr5 is tetrahedral. This is incorrect because the trigonal bipyramidal geometry is the most stable arrangement for a molecule with five atoms around a central atom.
📝 Note: Always double-check the number of valence electrons and the arrangement of atoms when drawing the Lewis structure to avoid common misconceptions.
Visualizing the Pbr5 Lewis Structure
Visualizing the Pbr5 Lewis Structure can be challenging, but it is essential for understanding the molecular geometry and bonding. Here is an image that illustrates the Lewis structure and molecular geometry of PBr5:

This image shows the trigonal bipyramidal geometry of PBr5, with the phosphorus atom at the center and the five bromine atoms at the vertices. The single bonds between the phosphorus and bromine atoms are clearly visible, and the lone pairs on the bromine atoms are not shown for simplicity.
Understanding the Pbr5 Lewis Structure is crucial for anyone studying chemistry. It provides insights into the molecular geometry, bonding, and reactivity of PBr5. By following the steps outlined in this post, you can draw an accurate Lewis structure for PBr5 and understand its molecular properties. The trigonal bipyramidal geometry of PBr5 is a result of the arrangement of atoms and electrons, which minimizes electron repulsion and provides stability to the molecule. This knowledge is essential for predicting the chemical properties and reactivity of PBr5 and designing experiments for further study.
Related Terms:
- pbr5 compound name
- pbr5 polar or nonpolar
- pbr5 electron geometry
- pbr5 diagram
- pbr5 hybridization
- molecular geometry of pbr5