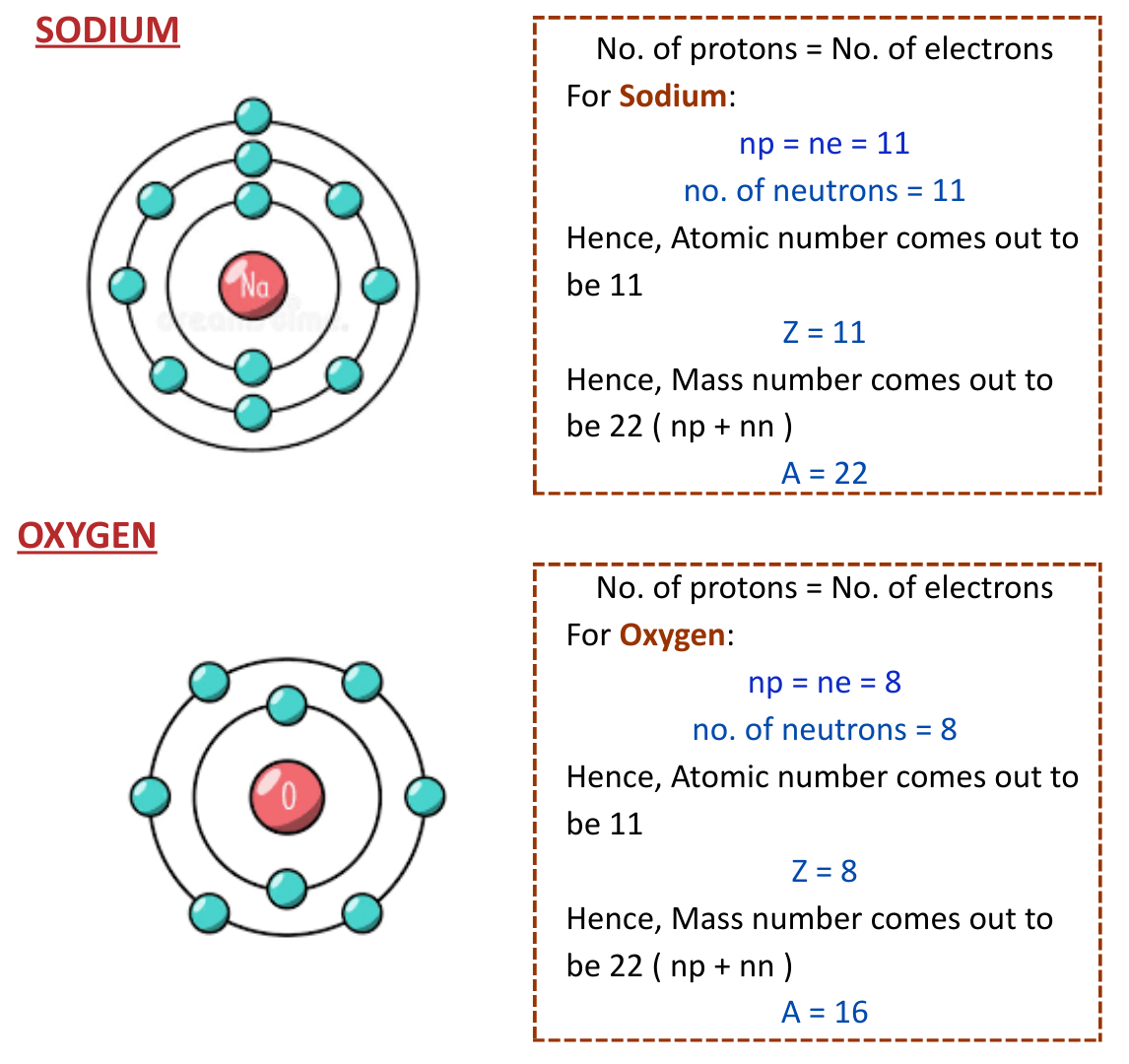
Oxygen is a fundamental element in the periodic table, essential for life on Earth. Understanding its atomic structure, including the oxygen number of neutrons, is crucial for various scientific and industrial applications. This blog post delves into the atomic composition of oxygen, its isotopes, and the significance of the oxygen number of neutrons in different contexts.
Understanding the Atomic Structure of Oxygen
Oxygen, with the atomic number 8, has a total of 8 protons in its nucleus. The number of neutrons in an oxygen atom can vary, leading to different isotopes. The most common isotope of oxygen is 16O, which has 8 neutrons. This makes the oxygen number of neutrons in 16O equal to 8.
The Importance of Oxygen Isotopes
Oxygen has several isotopes, each with a different number of neutrons. The most abundant isotopes are:
| Isotope | Number of Neutrons | Abundance |
|---|---|---|
| 16O | 8 | 99.762% |
| 17O | 9 | 0.038% |
| 18O | 10 | 0.200% |
These isotopes play crucial roles in various scientific fields, including geology, climatology, and biology. The oxygen number of neutrons in these isotopes affects their stability and reactivity, making them valuable for different applications.
Applications of Oxygen Isotopes
Oxygen isotopes are used in a wide range of applications due to their unique properties. Some of the key applications include:
- Geology and Paleoclimatology: The ratio of 18O to 16O in geological samples can provide insights into past climate conditions. This is because the oxygen number of neutrons affects the isotope’s behavior in different temperature and pressure conditions.
- Medicine: Oxygen isotopes, particularly 18O, are used in medical imaging techniques such as Positron Emission Tomography (PET) scans. The oxygen number of neutrons in 18O makes it suitable for labeling compounds used in these scans.
- Environmental Science: Oxygen isotopes are used to study water cycles and environmental changes. The oxygen number of neutrons in different isotopes helps track the movement of water molecules through various ecosystems.
The Role of Oxygen in Biological Systems
Oxygen is vital for most life forms on Earth. It plays a crucial role in cellular respiration, where it helps convert nutrients into energy. The oxygen number of neutrons in the most common isotope, 16O, ensures its stability and reactivity, making it ideal for biological processes.
In biological systems, oxygen is often transported by hemoglobin in red blood cells. The oxygen number of neutrons in the oxygen molecules bound to hemoglobin affects their affinity for oxygen, influencing how efficiently oxygen is delivered to tissues.
Oxygen in Industrial Applications
Oxygen is widely used in various industrial processes. Its reactivity, influenced by the oxygen number of neutrons, makes it a valuable component in many chemical reactions. Some key industrial applications include:
- Steel Production: Oxygen is used to remove impurities from molten steel, improving its quality. The oxygen number of neutrons in the oxygen molecules affects their reactivity with impurities.
- Welding and Cutting: Oxygen is used in welding and cutting processes to create high-temperature flames. The oxygen number of neutrons in the oxygen molecules ensures efficient combustion.
- Chemical Synthesis: Oxygen is a key reactant in many chemical synthesis processes. The oxygen number of neutrons in the oxygen molecules influences their reactivity with other compounds.
📝 Note: The oxygen number of neutrons can vary depending on the isotope, affecting its stability and reactivity. Understanding these variations is crucial for optimizing the use of oxygen in different applications.
Oxygen in Environmental Science
Oxygen plays a critical role in environmental science, particularly in studying climate change and water cycles. The oxygen number of neutrons in different isotopes helps scientists track the movement of water molecules and understand past climate conditions.
For example, the ratio of 18O to 16O in ice cores can provide insights into temperature changes over thousands of years. The oxygen number of neutrons in these isotopes affects their behavior in different temperature and pressure conditions, making them valuable for climate research.
Future Directions in Oxygen Research
Research on oxygen and its isotopes continues to evolve, with new applications and discoveries being made regularly. The oxygen number of neutrons remains a key focus, as it influences the stability and reactivity of different isotopes. Future research may explore:
- New Isotopic Applications: Discovering new uses for oxygen isotopes in medicine, environmental science, and industry.
- Advanced Analytical Techniques: Developing more sensitive and accurate methods for measuring oxygen isotopes.
- Climate Modeling: Using oxygen isotopes to improve climate models and predict future environmental changes.
As our understanding of oxygen and its isotopes deepens, so too will our ability to harness their unique properties for the benefit of society.
In summary, oxygen is a versatile element with a rich atomic structure. The oxygen number of neutrons plays a crucial role in determining its properties and applications. From biological systems to industrial processes and environmental science, oxygen’s unique characteristics make it indispensable. As research continues, we can expect to uncover even more about this essential element and its isotopes, paving the way for new discoveries and innovations.
Related Terms:
- potassium number of neutrons
- oxygen number of protons
- oxygen atomic number
- hydrogen number of neutrons
- nitrogen number of neutrons
- fluorine number of neutrons