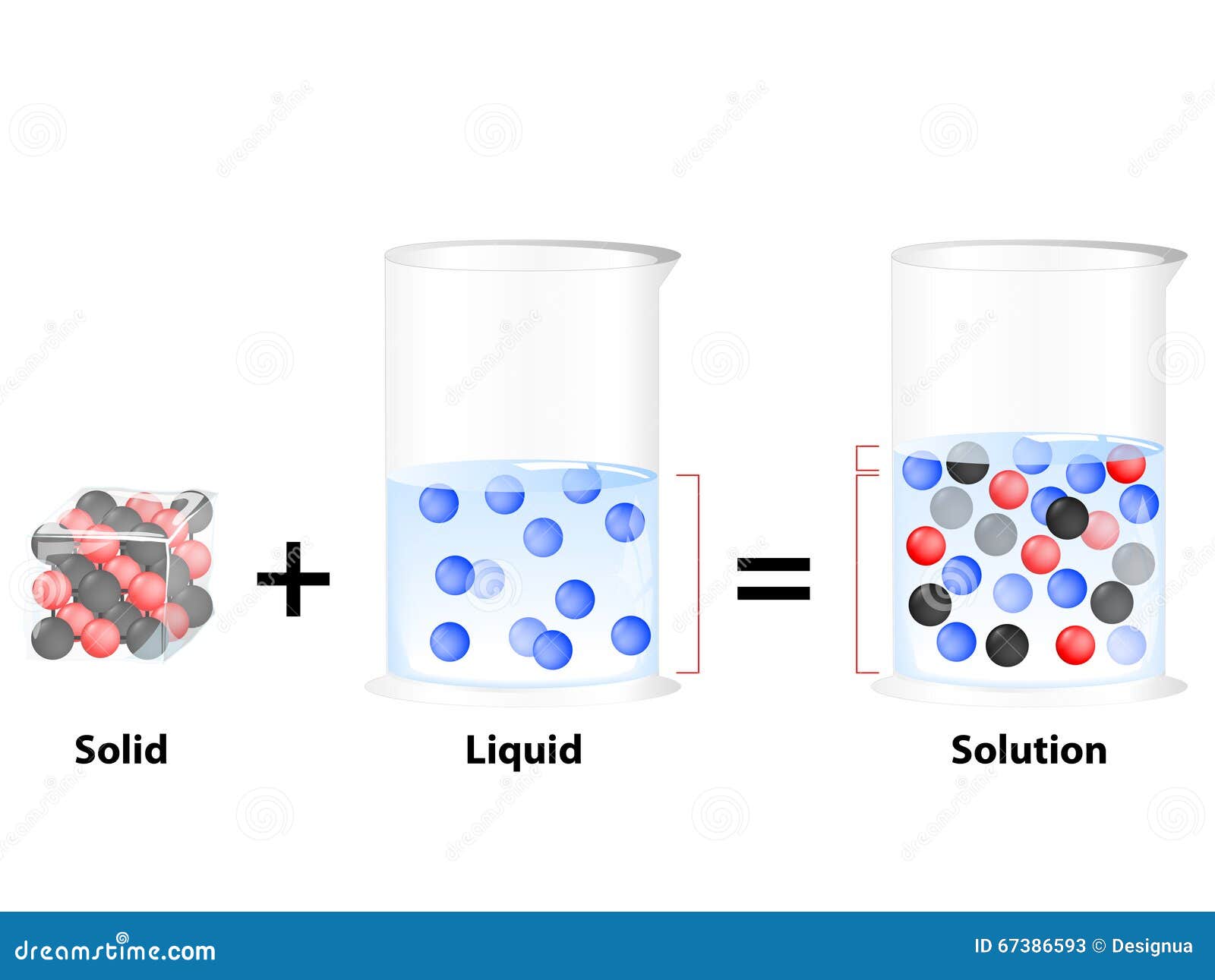
Understanding the distinction between a mixture and a solution is fundamental in chemistry and various scientific disciplines. Both terms refer to combinations of substances, but they differ in their properties and behaviors. This blog post will delve into the definitions, characteristics, and applications of mixtures and solutions, providing a comprehensive overview to help you grasp these essential concepts.
What is a Mixture?
A mixture is a combination of two or more substances that are not chemically bonded together. The substances in a mixture retain their individual properties and can be separated by physical means. Mixtures can be either homogeneous or heterogeneous.
Types of Mixtures
Mixtures can be categorized into two main types based on their homogeneity:
- Homogeneous Mixtures: These mixtures have a uniform composition throughout. Examples include air, saltwater, and alloys like brass.
- Heterogeneous Mixtures: These mixtures do not have a uniform composition. Examples include sand and water, oil and water, and a salad.
Characteristics of Mixtures
Mixtures have several key characteristics:
- Variable Composition: The components of a mixture can vary in proportion.
- Physical Separation: The components can be separated by physical methods such as filtration, distillation, or evaporation.
- No Chemical Reaction: The components do not undergo a chemical reaction when mixed.
What is a Solution?
A solution is a type of homogeneous mixture where one substance (the solute) is dissolved in another substance (the solvent). Solutions are characterized by the uniform distribution of the solute particles within the solvent. Solutions can be solid, liquid, or gaseous.
Components of a Solution
A solution consists of two main components:
- Solute: The substance that is dissolved in the solvent. It can be a solid, liquid, or gas.
- Solvent: The substance that dissolves the solute. It is usually the component present in the larger amount.
Types of Solutions
Solutions can be classified based on the state of the solute and solvent:
- Solid Solutions: Examples include alloys like brass (copper and zinc) and amalgams (mercury and other metals).
- Liquid Solutions: Examples include saltwater (sodium chloride in water) and alcohol in water.
- Gaseous Solutions: Examples include air (a mixture of gases like nitrogen, oxygen, and carbon dioxide).
Characteristics of Solutions
Solutions have several key characteristics:
- Uniform Composition: The solute is evenly distributed throughout the solvent.
- Stability: The components do not separate over time.
- No Chemical Reaction: The solute and solvent do not undergo a chemical reaction when mixed.
Differences Between Mixtures and Solutions
While both mixtures and solutions involve the combination of substances, there are distinct differences between them. Here is a comparison:
| Characteristic | Mixture | Solution |
|---|---|---|
| Homogeneity | Can be homogeneous or heterogeneous | Always homogeneous |
| Composition | Variable | Uniform |
| Separation | Can be separated by physical means | Cannot be separated by physical means |
| Chemical Reaction | No chemical reaction | No chemical reaction |
💡 Note: The key difference lies in the homogeneity and uniformity of the components. Solutions are always homogeneous, while mixtures can be either homogeneous or heterogeneous.
Applications of Mixtures and Solutions
Mixtures and solutions have wide-ranging applications in various fields, including chemistry, medicine, and industry. Here are some examples:
Applications of Mixtures
- Alloys: Mixtures of metals used in construction and manufacturing, such as steel (iron and carbon) and brass (copper and zinc).
- Concrete: A mixture of cement, sand, and gravel used in construction.
- Pharmaceuticals: Mixtures of active ingredients and excipients in medications.
Applications of Solutions
- Medicines: Solutions like saline (sodium chloride in water) used for intravenous fluids.
- Cleaning Agents: Solutions like bleach (sodium hypochlorite in water) used for disinfection.
- Food and Beverages: Solutions like sugar water (sucrose in water) used in beverages.
Preparing a Mixture or Solution
Preparing a mixture or solution involves combining the components in the desired proportions. Here are the steps for preparing both:
Preparing a Mixture
To prepare a mixture, follow these steps:
- Gather the components you want to mix.
- Combine the components in the desired proportions.
- Mix thoroughly to ensure homogeneity if desired.
- Store the mixture in an appropriate container.
💡 Note: The method of mixing can vary depending on the type of mixture. For example, a heterogeneous mixture like sand and water can be mixed by stirring, while a homogeneous mixture like air can be mixed by diffusion.
Preparing a Solution
To prepare a solution, follow these steps:
- Gather the solute and solvent.
- Measure the desired amount of solute and solvent.
- Add the solute to the solvent and stir until the solute is completely dissolved.
- Store the solution in an appropriate container.
💡 Note: The solubility of the solute in the solvent can affect the preparation process. Some solutes may require heating or agitation to dissolve completely.
Examples of Mixtures and Solutions in Daily Life
Mixtures and solutions are ubiquitous in our daily lives. Here are some examples:
Examples of Mixtures
- Air: A mixture of gases like nitrogen, oxygen, and carbon dioxide.
- Soil: A mixture of minerals, organic matter, and water.
- Trail Mix: A mixture of nuts, dried fruits, and chocolate chips.
Examples of Solutions
- Soft Drinks: Solutions of sugar, flavorings, and carbon dioxide in water.
- Vinegar: A solution of acetic acid in water.
- Hand Sanitizer: A solution of alcohol in water with added fragrances and moisturizers.
Understanding the distinction between mixtures and solutions is crucial for various applications in science, industry, and daily life. By recognizing the characteristics and behaviors of these combinations, you can better appreciate their roles in different contexts and utilize them effectively.
Related Terms:
- mixture and solution examples
- examples of mixtures and solutions
- solution vs mixture chemistry
- mixture or solution worksheet
- solution vs heterogeneous mixture
- mixture vs solution examples