Understanding the Li Electron Config is fundamental for anyone delving into the world of chemistry, particularly in the study of atomic structure and periodic trends. Lithium, with the atomic number 3, is the third element in the periodic table and the first in the alkali metal group. Its electron configuration provides insights into its chemical behavior and reactivity. This post will explore the Li Electron Config, its significance, and how it influences the properties of lithium.
What is Electron Configuration?
Electron configuration refers to the distribution of electrons in the various orbitals of an atom. It is a crucial concept in understanding the chemical properties of elements. The configuration is typically written using a notation that specifies the energy levels (n) and the type of orbital (s, p, d, f) along with the number of electrons in each orbital.
Understanding the Li Electron Config
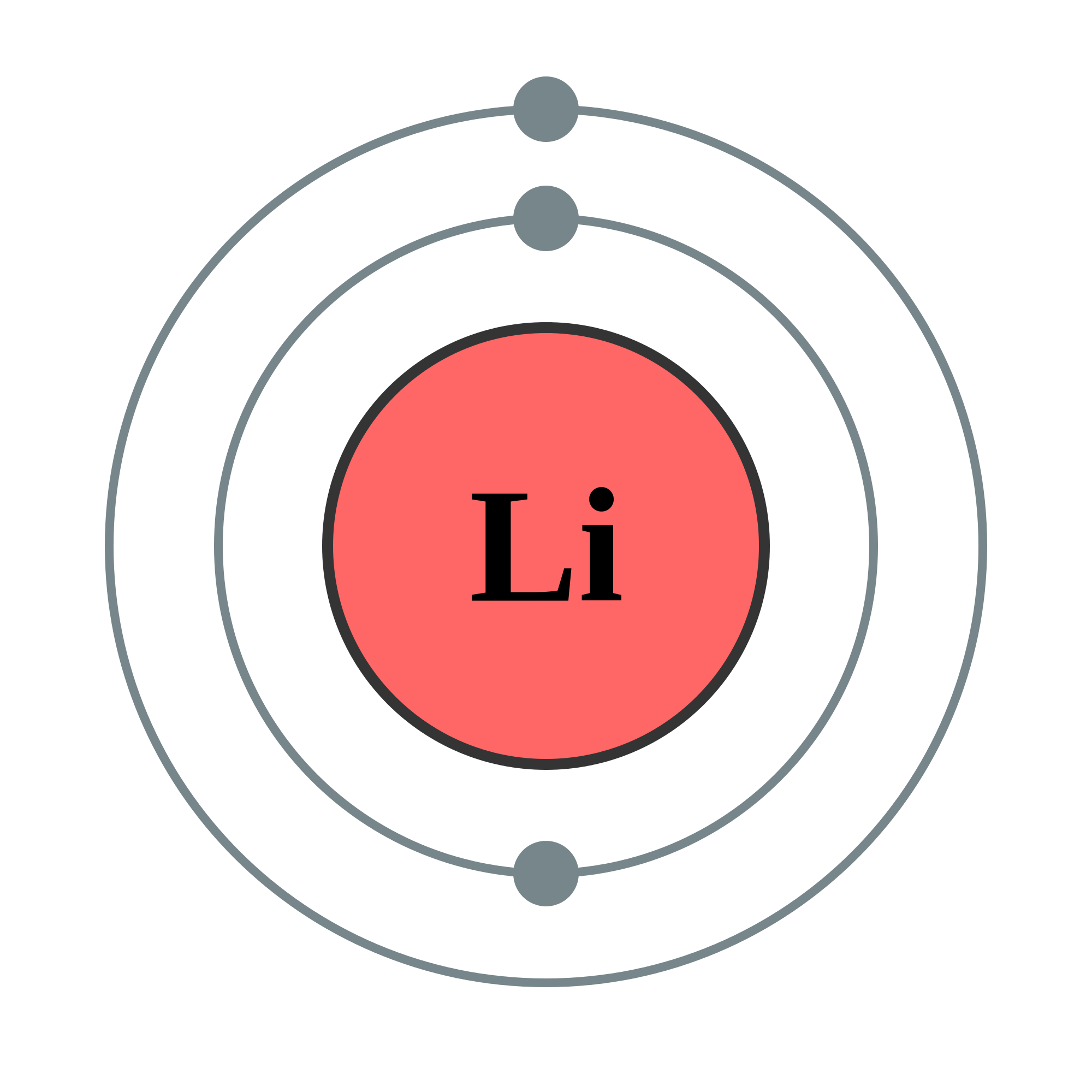
The Li Electron Config can be determined by following the Aufbau principle, which states that electrons fill the lowest energy orbitals first. Lithium has three electrons, and its electron configuration is written as:
1s22s1
This notation indicates that:
- The first two electrons fill the 1s orbital.
- The third electron occupies the 2s orbital.
This configuration is essential for understanding lithium’s chemical behavior and its position in the periodic table.
Significance of the Li Electron Config
The Li Electron Config has several significant implications:
- Valence Electrons: Lithium has one valence electron in its outermost shell (2s1). This single electron in the outermost shell makes lithium highly reactive, as it tends to lose this electron to achieve a stable noble gas configuration.
- Chemical Reactivity: The presence of a single valence electron makes lithium highly reactive. It readily forms compounds with non-metals, such as lithium oxide (Li2O) and lithium chloride (LiCl).
- Alkali Metal Properties: Lithium’s electron configuration places it in the alkali metal group, which includes elements like sodium (Na) and potassium (K). These elements share similar properties, such as low ionization energies and high reactivity.
Periodic Trends and the Li Electron Config
The Li Electron Config also helps in understanding periodic trends. As you move from left to right across a period in the periodic table, the number of electrons in the outermost shell increases. This increase affects the atomic radius, ionization energy, and electronegativity of the elements.
For lithium, being the first element in its period, it has a relatively large atomic radius and low ionization energy compared to other elements in the same period. This is because it has only one electron in its outermost shell, which is easily removed.
Comparing Li Electron Config with Other Alkali Metals
To better understand the Li Electron Config, it is helpful to compare it with the electron configurations of other alkali metals. The table below shows the electron configurations of the first few alkali metals:
| Element | Atomic Number | Electron Configuration |
|---|---|---|
| Lithium (Li) | 3 | 1s22s1 |
| Sodium (Na) | 11 | 1s22s22p63s1 |
| Potassium (K) | 19 | 1s22s22p63s23p64s1 |
| Rubidium (Rb) | 37 | 1s22s22p63s23p63d104s24p65s1 |
| Cesium (Cs) | 55 | 1s22s22p63s23p63d104s24p64d105s25p66s1 |
As seen in the table, all alkali metals have a single electron in their outermost s orbital, which is responsible for their similar chemical properties. The number of inner electrons increases as you move down the group, leading to larger atomic radii and lower ionization energies.
Applications of Lithium
The unique Li Electron Config makes lithium a valuable element in various applications. Some of the key uses of lithium include:
- Batteries: Lithium is widely used in rechargeable batteries, such as lithium-ion batteries, which power many electronic devices, including smartphones, laptops, and electric vehicles.
- Medicine: Lithium compounds, such as lithium carbonate, are used in the treatment of bipolar disorder and other mental health conditions.
- Alloys: Lithium is used in the production of alloys with other metals, such as aluminum and magnesium, to improve their strength and durability.
- Lubricants: Lithium greases are used as high-performance lubricants in various industrial applications.
Safety and Handling of Lithium
While lithium has many beneficial applications, it is also highly reactive and must be handled with care. Some safety considerations include:
- Reactivity: Lithium reacts vigorously with water, producing hydrogen gas and lithium hydroxide. This reaction can be explosive, so lithium should be kept away from water and moisture.
- Flammability: Lithium is highly flammable and can ignite spontaneously in air. It should be stored in a dry, inert atmosphere to prevent combustion.
- Toxicity: Lithium compounds can be toxic if ingested or inhaled. Proper personal protective equipment (PPE) should be used when handling lithium.
🛑 Note: Always follow safety protocols and guidelines when handling lithium to prevent accidents and ensure safe use.
Future Prospects of Lithium
The demand for lithium is expected to grow significantly in the coming years, driven by the increasing use of lithium-ion batteries in electric vehicles and energy storage systems. This growth presents both opportunities and challenges for the lithium industry. Some key areas of focus include:
- Sustainable Mining: Developing sustainable and environmentally friendly methods for lithium mining and extraction.
- Recycling: Improving the recycling of lithium-ion batteries to reduce waste and conserve resources.
- Innovation: Researching and developing new lithium-based materials and technologies to enhance performance and efficiency.
In conclusion, the Li Electron Config is a fundamental concept that underpins the chemical behavior and properties of lithium. Understanding this configuration provides insights into lithium’s reactivity, its position in the periodic table, and its various applications. As the demand for lithium continues to grow, so too will the importance of understanding and optimizing its electron configuration for future technological advancements. The unique properties of lithium, driven by its electron configuration, make it a crucial element in modern technology and industry, with a bright future ahead.
Related Terms:
- electron config of lithium
- li valence electrons
- li 1 electron configuration
- full electron configuration for lithium
- the electron configuration of lithium
- lithium electron configuration chart