Understanding the Lewis structure of molecules is fundamental in chemistry, as it helps visualize the arrangement of valence electrons around atoms. One such molecule that often intrigues students and chemists alike is sulfur tetrafluoride (SF4). This molecule has a unique structure that deviates from the typical tetrahedral geometry seen in many other molecules. In this post, we will delve into the Lewis structure of SF4, its molecular geometry, and the underlying principles that govern its formation.
Understanding Lewis Structures
Lewis structures, also known as Lewis dot diagrams, are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. They are named after Gilbert N. Lewis, who introduced the concept in 1916. The basic rules for drawing Lewis structures include:
- Determine the total number of valence electrons in the molecule.
- Identify the central atom, which is usually the least electronegative element.
- Arrange the other atoms around the central atom.
- Distribute the valence electrons to form bonds and satisfy the octet rule.
- Place any remaining electrons as lone pairs on the atoms.
The Lewis Structure of SF4
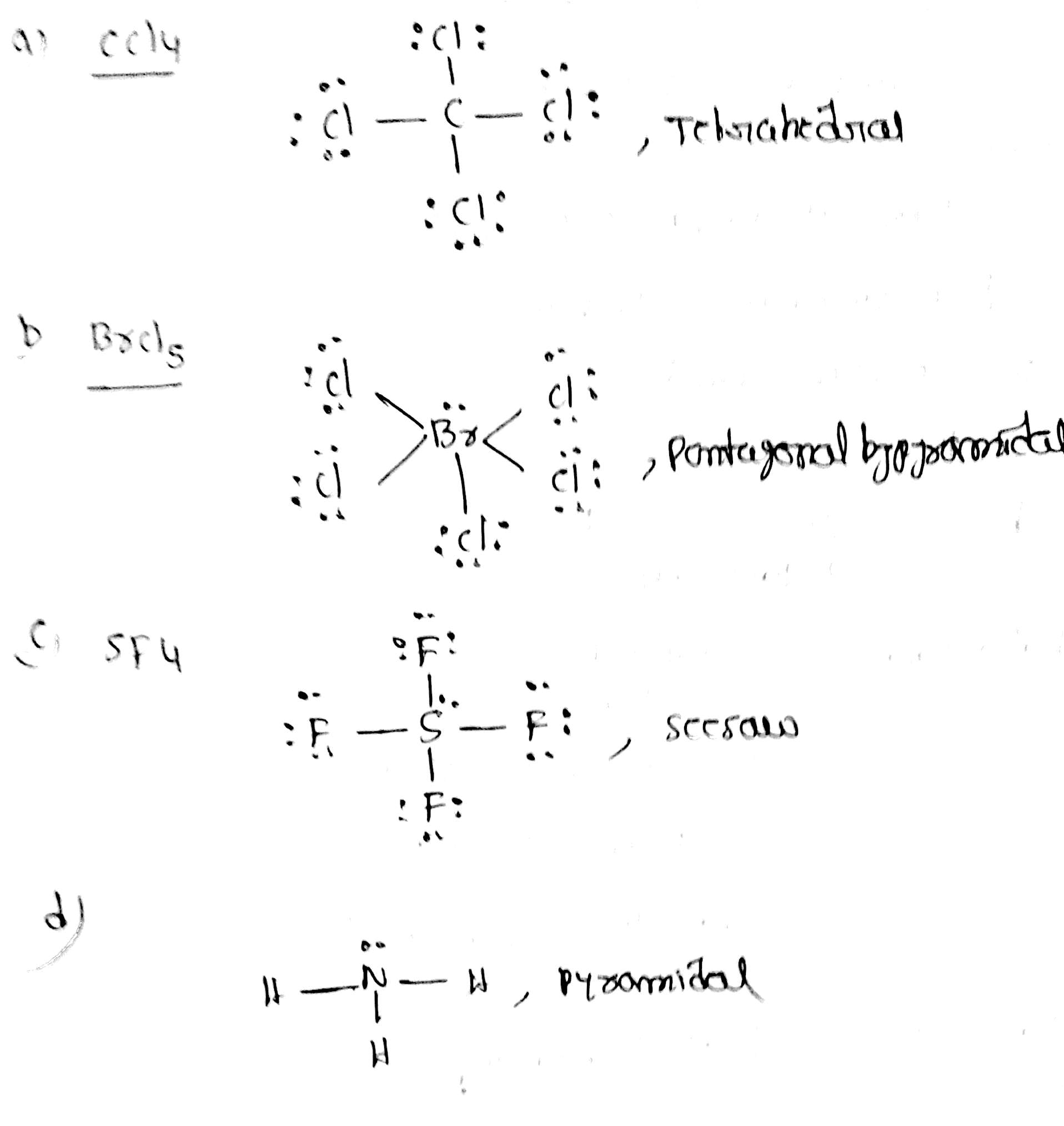
Sulfur tetrafluoride (SF4) consists of one sulfur atom and four fluorine atoms. Sulfur is the central atom because it is less electronegative than fluorine. Let’s go through the steps to draw the Lewis structure of SF4.
Step 1: Determine the Total Number of Valence Electrons
Sulfur (S) is in group 16 of the periodic table and has 6 valence electrons. Fluorine (F) is in group 17 and has 7 valence electrons. Since there are four fluorine atoms, the total number of valence electrons is:
6 (from S) + 4 × 7 (from F) = 6 + 28 = 34 valence electrons.
Step 2: Arrange the Atoms
Place the sulfur atom in the center and the four fluorine atoms around it.
Step 3: Form Single Bonds
Connect each fluorine atom to the sulfur atom with a single bond. This uses up 8 electrons (4 bonds × 2 electrons per bond).
Step 4: Distribute Remaining Electrons
After forming the single bonds, we have 34 - 8 = 26 electrons left. These electrons are distributed as lone pairs on the fluorine atoms. Each fluorine atom needs 3 lone pairs to complete its octet, which accounts for 24 electrons. This leaves 2 electrons, which are placed as a lone pair on the sulfur atom.
Step 5: Check the Octet Rule
Each fluorine atom has 8 electrons (1 bond pair + 3 lone pairs), satisfying the octet rule. The sulfur atom has 10 electrons (4 bond pairs + 1 lone pair), which is an exception to the octet rule but is common for sulfur due to its ability to expand its valence shell.
Molecular Geometry of SF4
The molecular geometry of SF4 is not straightforward due to the presence of the lone pair on the sulfur atom. According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, the geometry is determined by the repulsion between electron pairs. In SF4, there are five electron pairs around the sulfur atom (four bond pairs and one lone pair).
The ideal geometry for five electron pairs is a trigonal bipyramidal arrangement. However, the presence of the lone pair distorts this geometry. The lone pair repels the bond pairs more strongly, leading to a seesaw shape. In this shape, two fluorine atoms are in axial positions, and the other two are in equatorial positions. The axial fluorine atoms are at a 173° angle to each other, while the equatorial fluorine atoms are at a 120° angle to each other.
Importance of the Lewis Structure of SF4
The Lewis structure of SF4 is crucial for understanding its chemical properties and reactivity. The presence of the lone pair on the sulfur atom makes SF4 a polar molecule, despite the non-polar nature of the individual S-F bonds. This polarity affects its solubility, reactivity, and other physical properties.
Additionally, the Lewis structure helps in predicting the behavior of SF4 in chemical reactions. For example, the lone pair on the sulfur atom can act as a nucleophile, attacking electrophilic centers in other molecules. This reactivity is essential in various chemical processes, including catalysis and synthesis.
Comparing SF4 with Other Sulfur Fluorides
Sulfur forms several fluorides, each with its unique Lewis structure and properties. Comparing SF4 with other sulfur fluorides provides insights into the trends and patterns in their chemistry.
| Molecule | Lewis Structure | Molecular Geometry | Polarity |
|---|---|---|---|
| SF2 | S has 2 bond pairs and 2 lone pairs | Bent | Polar |
| SF4 | S has 4 bond pairs and 1 lone pair | Seesaw | Polar |
| SF6 | S has 6 bond pairs | Octahedral | Non-polar |
As seen in the table, the number of fluorine atoms and the resulting electron pairs around the sulfur atom significantly influence the molecular geometry and polarity. SF2 and SF4 are polar due to the presence of lone pairs, while SF6 is non-polar because it has no lone pairs and a symmetrical octahedral geometry.
💡 Note: The polarity of a molecule is determined by the net dipole moment, which is the vector sum of the individual bond dipoles. In SF4, the lone pair on the sulfur atom creates a significant dipole moment, making the molecule polar.
Applications of SF4
Sulfur tetrafluoride has several applications in industry and research. Its reactivity and unique properties make it a valuable compound in various fields.
One of the primary applications of SF4 is in the synthesis of organofluorine compounds. These compounds are used in pharmaceuticals, agrochemicals, and materials science. The ability of SF4 to introduce fluorine atoms into organic molecules makes it a useful reagent in synthetic chemistry.
Additionally, SF4 is used in the production of certain types of glass and ceramics. Its reactivity with silicon dioxide (SiO2) helps in the formation of fluorosilicate glasses, which have unique optical and thermal properties.
In research, SF4 is studied for its potential applications in catalysis and materials science. Its ability to form stable complexes with various metals makes it a promising candidate for developing new catalytic systems.
SF4 is also used in the semiconductor industry for etching and cleaning processes. Its reactivity with silicon and other materials makes it effective in removing unwanted layers and contaminants from semiconductor surfaces.
Despite its usefulness, handling SF4 requires caution due to its toxicity and reactivity. Proper safety measures, including the use of personal protective equipment and adequate ventilation, are essential when working with this compound.
In summary, the Lewis structure of SF4 provides valuable insights into its molecular geometry, polarity, and reactivity. Understanding these aspects is crucial for utilizing SF4 in various applications and for predicting its behavior in chemical reactions. The unique properties of SF4 make it a fascinating molecule to study and a useful compound in industry and research.
Related Terms:
- shape of sf4
- lewis structure of sf2
- sf4 molecular shape
- lewis structure of ch4
- molecular structure of sf4
- molecular geometry of sf4