Understanding the differences between ionic and molecular compounds is fundamental in chemistry. These two types of compounds exhibit distinct properties and behaviors due to their unique structures and bonding mechanisms. This blog post delves into the Ionic Vs Molecular comparison, exploring their definitions, properties, and applications.
What are Ionic Compounds?
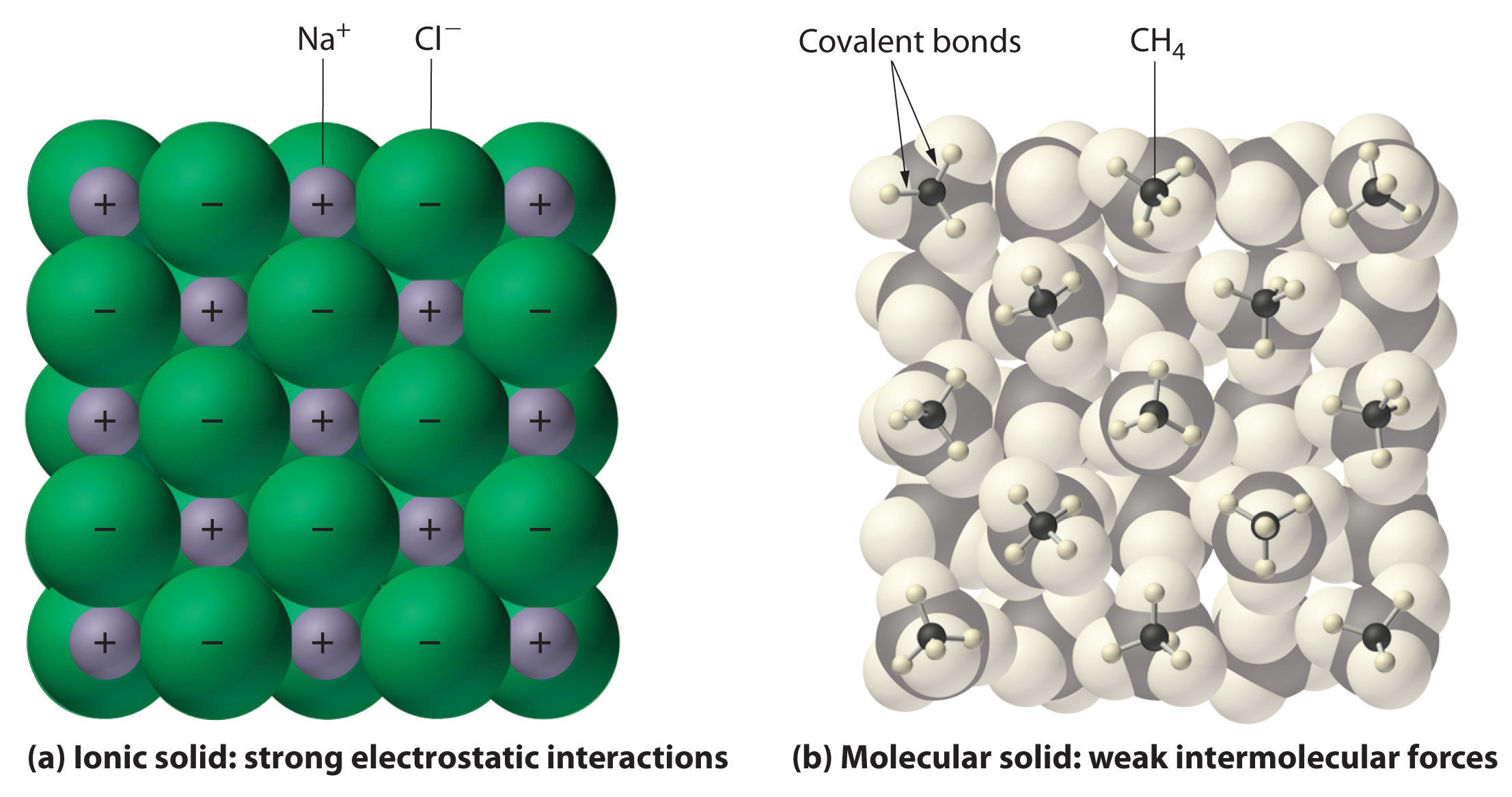
Ionic compounds are formed through the transfer of electrons between atoms, resulting in the creation of ions. These ions are held together by strong electrostatic forces known as ionic bonds. The key characteristic of ionic compounds is that they consist of a metal and a non-metal.
For example, sodium chloride (NaCl), commonly known as table salt, is an ionic compound. In this compound, sodium (Na) loses an electron to become a positively charged ion (Na+), while chlorine (Cl) gains an electron to become a negatively charged ion (Cl-). The strong attraction between these oppositely charged ions forms the ionic bond.
Properties of Ionic Compounds
Ionic compounds have several distinctive properties:
- High Melting and Boiling Points: Due to the strong electrostatic forces between ions, ionic compounds typically have high melting and boiling points.
- Conductivity: In their molten or dissolved state, ionic compounds conduct electricity because the ions are free to move and carry electrical charge.
- Solubility: Many ionic compounds are soluble in water, forming solutions that conduct electricity.
- Crystal Structure: Ionic compounds often form crystalline structures, where ions are arranged in a regular, repeating pattern.
What are Molecular Compounds?
Molecular compounds, on the other hand, are formed through the sharing of electrons between atoms. This sharing results in covalent bonds, which hold the atoms together within a molecule. Molecular compounds are typically composed of non-metals.
For instance, water (H2O) is a molecular compound. In a water molecule, two hydrogen atoms share electrons with one oxygen atom, forming covalent bonds. The shared electrons create a stable molecular structure.
Properties of Molecular Compounds
Molecular compounds exhibit different properties compared to ionic compounds:
- Lower Melting and Boiling Points: Molecular compounds generally have lower melting and boiling points because the forces between molecules (intermolecular forces) are weaker than ionic bonds.
- Non-Conductivity: Molecular compounds do not conduct electricity in their solid, liquid, or dissolved states because they do not contain free ions or electrons.
- Solubility: The solubility of molecular compounds varies widely. Some are soluble in water, while others are not. Solubility depends on the polarity of the molecule and the solvent.
- Physical State: Molecular compounds can exist in various physical states—solid, liquid, or gas—at room temperature.
Ionic Vs Molecular: A Comparative Analysis
To better understand the differences between ionic and molecular compounds, let's compare them side by side:
| Property | Ionic Compounds | Molecular Compounds |
|---|---|---|
| Bonding | Ionic bonds (electron transfer) | Covalent bonds (electron sharing) |
| Composition | Metal and non-metal | Non-metals |
| Melting and Boiling Points | High | Low |
| Conductivity | Conduct in molten or dissolved state | Do not conduct |
| Solubility | Many are soluble in water | Varies widely |
| Crystal Structure | Often form crystals | Can be solid, liquid, or gas |
This comparison highlights the fundamental differences between ionic and molecular compounds, emphasizing their unique characteristics and behaviors.
Applications of Ionic and Molecular Compounds
Both ionic and molecular compounds have numerous applications in various fields. Understanding their properties is crucial for their effective use.
Applications of Ionic Compounds
Ionic compounds are widely used in:
- Medicine: Many ionic compounds, such as sodium chloride and potassium chloride, are essential for maintaining bodily functions.
- Industry: Ionic compounds like sodium hydroxide (NaOH) are used in the production of soaps, detergents, and paper.
- Agriculture: Fertilizers often contain ionic compounds like ammonium nitrate (NH4NO3) to provide essential nutrients to plants.
Applications of Molecular Compounds
Molecular compounds are used in:
- Pharmaceuticals: Many drugs, such as aspirin and paracetamol, are molecular compounds.
- Chemical Industry: Molecular compounds like methanol (CH3OH) and ethanol (C2H5OH) are used as solvents and fuels.
- Food Industry: Molecular compounds like sucrose (table sugar) and fructose are essential ingredients in food and beverages.
💡 Note: The applications of ionic and molecular compounds are vast and varied, reflecting their diverse properties and uses in different industries.
Conclusion
In summary, ionic and molecular compounds differ significantly in their bonding mechanisms, properties, and applications. Ionic compounds, formed through electron transfer, exhibit high melting points, conductivity in molten or dissolved states, and often form crystalline structures. In contrast, molecular compounds, formed through electron sharing, have lower melting points, do not conduct electricity, and can exist in various physical states. Understanding these differences is crucial for their effective use in various fields, from medicine and industry to agriculture and beyond.
Related Terms:
- molecular vs ionic bonds
- molecular vs ionic examples
- difference between molecular and ionic
- ionic vs molecular compounds characteristics
- molecular vs ionic equation
- molecular vs ionic compound examples