Understanding the fundamental differences between Ionic vs Molecular Bonds is crucial for anyone delving into the world of chemistry. These two types of chemical bonds are the backbone of molecular structures and play pivotal roles in determining the properties of substances. This blog post will explore the distinctions between ionic and molecular bonds, their formation, properties, and examples to provide a comprehensive understanding.
What are Ionic Bonds?
Ionic bonds are formed through the transfer of electrons from one atom to another, resulting in the creation of ions. These ions are then attracted to each other due to electrostatic forces. The atom that loses electrons becomes a positively charged cation, while the atom that gains electrons becomes a negatively charged anion.
Formation of Ionic Bonds
Ionic bonds typically form between metals and non-metals. Metals tend to have low ionization energies, making it easier for them to lose electrons, while non-metals have high electron affinities, making it easier for them to gain electrons. The process can be broken down into the following steps:
- An atom of a metal loses one or more electrons to become a cation.
- An atom of a non-metal gains one or more electrons to become an anion.
- The oppositely charged ions are attracted to each other, forming an ionic bond.
Properties of Ionic Bonds
Ionic compounds exhibit several characteristic properties:
- High Melting and Boiling Points: The strong electrostatic forces between ions require a lot of energy to overcome, resulting in high melting and boiling points.
- Conductivity: In the molten state or when dissolved in water, ionic compounds conduct electricity due to the presence of free ions.
- Solubility: Many ionic compounds are soluble in water, forming solutions that conduct electricity.
- Brittleness: Ionic compounds are often brittle and can shatter when subjected to physical stress.
Examples of Ionic Bonds
Some common examples of ionic compounds include:
- Sodium Chloride (NaCl): Formed between sodium (Na) and chlorine (Cl).
- Magnesium Oxide (MgO): Formed between magnesium (Mg) and oxygen (O).
- Calcium Fluoride (CaF2): Formed between calcium (Ca) and fluorine (F).
What are Molecular Bonds?
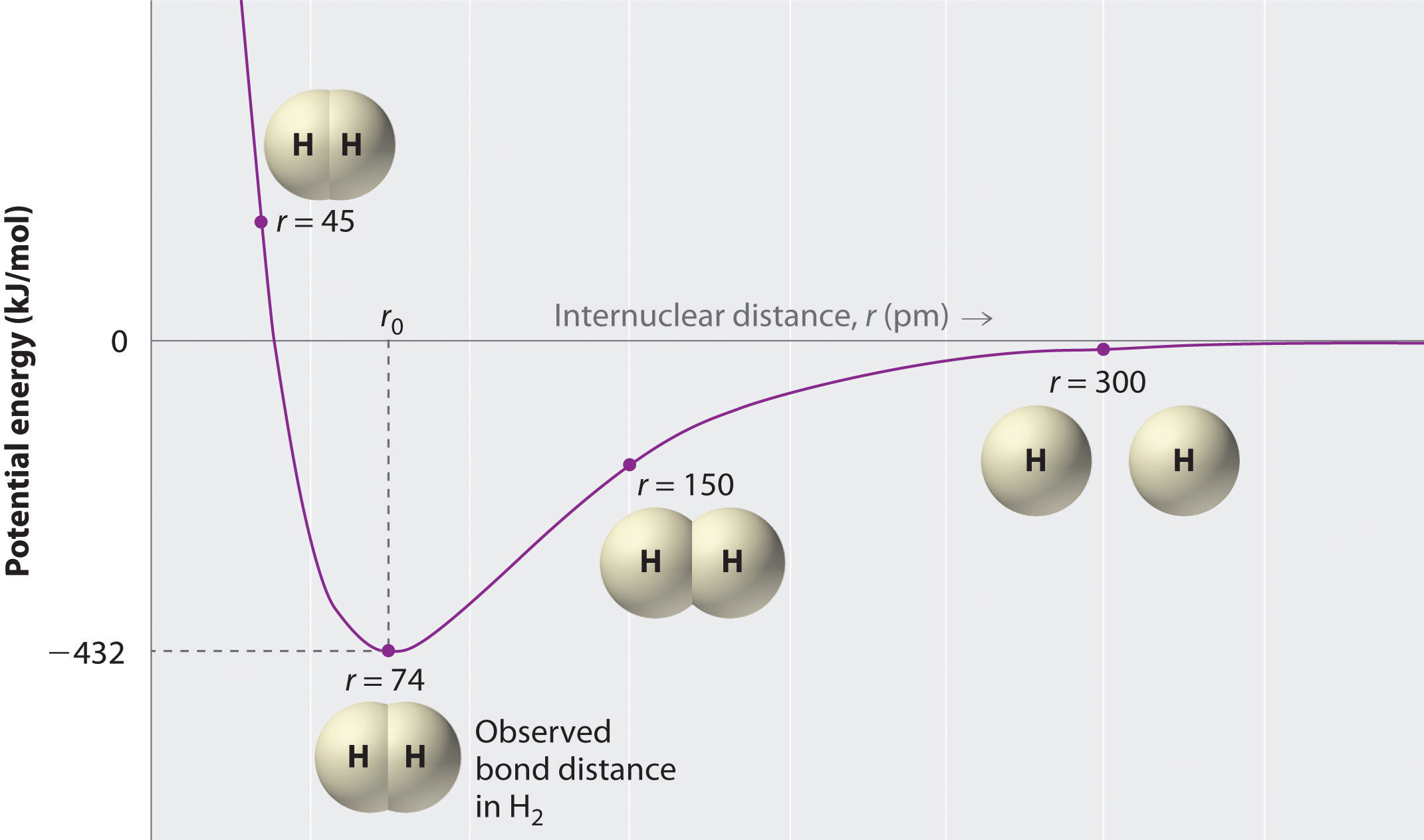
Molecular bonds, also known as covalent bonds, are formed when atoms share electrons to achieve a stable electron configuration. This sharing of electrons allows both atoms to fill their outer electron shells, resulting in a stable molecule.
Formation of Molecular Bonds
Molecular bonds typically form between non-metals. The process involves:
- Two or more atoms sharing one or more pairs of electrons.
- The shared electrons are attracted to the nuclei of both atoms, forming a covalent bond.
Properties of Molecular Bonds
Molecular compounds have distinct properties:
- Low Melting and Boiling Points: The intermolecular forces between molecules are generally weaker than the ionic bonds, resulting in lower melting and boiling points.
- Non-Conductivity: Molecular compounds do not conduct electricity in either the solid or liquid state because they do not have free ions or electrons.
- Solubility: Many molecular compounds are insoluble in water but soluble in organic solvents.
- Flexibility: Molecular compounds are often flexible and can exist in various states (solid, liquid, gas) under different conditions.
Examples of Molecular Bonds
Some common examples of molecular compounds include:
- Water (H2O): Formed between hydrogen (H) and oxygen (O).
- Methane (CH4): Formed between carbon (C) and hydrogen (H).
- Carbon Dioxide (CO2): Formed between carbon (C) and oxygen (O).
Ionic vs Molecular Bonds: A Comparative Analysis
To better understand the differences between Ionic vs Molecular Bonds, let's compare them across several key aspects:
| Aspect | Ionic Bonds | Molecular Bonds |
|---|---|---|
| Formation | Transfer of electrons between atoms | Sharing of electrons between atoms |
| Types of Atoms Involved | Metals and non-metals | Non-metals |
| Strength of Bond | Strong electrostatic forces | Weaker intermolecular forces |
| Melting and Boiling Points | High | Low |
| Conductivity | Conducts electricity in molten state or solution | Does not conduct electricity |
| Solubility | Many are soluble in water | Many are insoluble in water |
| Physical State | Often solid at room temperature | Can be solid, liquid, or gas at room temperature |
💡 Note: The table above provides a quick reference for the key differences between ionic and molecular bonds. Understanding these differences is essential for predicting the behavior and properties of various chemical compounds.
Applications of Ionic and Molecular Bonds
Both ionic and molecular bonds have wide-ranging applications in various fields. Understanding their properties and behaviors is crucial for their effective use in different industries.
Applications of Ionic Bonds
Ionic compounds are extensively used in:
- Medicine: Many ionic compounds are used in pharmaceuticals, such as sodium chloride (table salt) and potassium chloride (used in electrolyte solutions).
- Industry: Ionic compounds like sodium hydroxide (NaOH) are used in the production of soaps and detergents.
- Agriculture: Fertilizers often contain ionic compounds like ammonium nitrate (NH4NO3) and potassium sulfate (K2SO4).
Applications of Molecular Bonds
Molecular compounds are vital in:
- Chemistry: Many organic compounds, such as hydrocarbons and polymers, are based on molecular bonds.
- Biochemistry: Biological molecules like proteins, carbohydrates, and nucleic acids rely on molecular bonds for their structure and function.
- Environmental Science: Molecular compounds like carbon dioxide (CO2) and methane (CH4) play crucial roles in atmospheric chemistry and climate change.
Understanding the differences between Ionic vs Molecular Bonds is fundamental to grasping the behavior of chemical compounds and their applications in various fields. By recognizing the unique properties and formation mechanisms of these bonds, scientists and engineers can develop new materials, medicines, and technologies that improve our daily lives.
In summary, ionic bonds are formed through the transfer of electrons between metals and non-metals, resulting in strong electrostatic forces and high melting points. In contrast, molecular bonds involve the sharing of electrons between non-metals, leading to weaker intermolecular forces and lower melting points. Both types of bonds have distinct properties and applications, making them essential components of the chemical world. By studying these bonds, we gain a deeper understanding of the fundamental principles that govern chemical reactions and the behavior of matter.
Related Terms:
- how to find ionic compound
- identify ionic vs molecular compounds
- difference between molecule and ion
- how to determine ionic compounds
- different between ionic and molecular
- ionic vs molecular formula