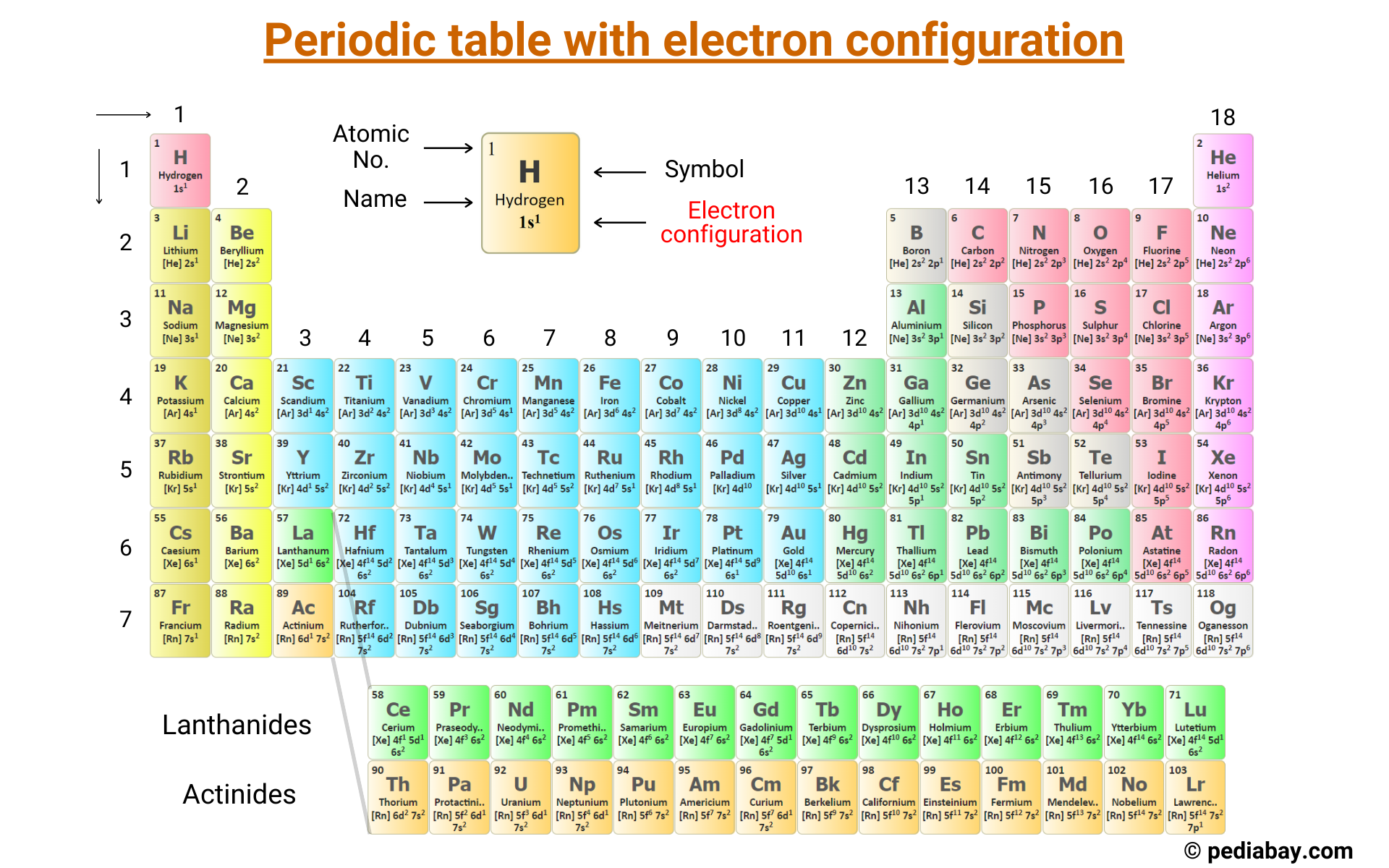
Understanding the electronic configuration of elements is fundamental to grasping the behavior and properties of matter at the atomic level. The electronic configuration, often abbreviated as Electronic Configuration Sc, refers to the distribution of electrons in the orbitals of an atom. This configuration is crucial for predicting chemical reactivity, bonding, and the physical properties of elements. In this post, we will delve into the intricacies of electronic configuration, focusing on the element scandium (Sc) as a case study.
What is Electronic Configuration?
Electronic configuration is the arrangement of electrons in the various orbitals of an atom. Each orbital can hold a specific number of electrons, and the distribution of these electrons determines the chemical properties of the element. The configuration is typically written using a notation that specifies the principal quantum number (n), the azimuthal quantum number (l), and the number of electrons in each orbital.
Understanding the Aufbau Principle
The Aufbau principle, also known as the building-up principle, states that electrons fill the lowest energy orbitals first before moving to higher energy levels. This principle is essential for determining the electronic configuration of any element. The order of filling orbitals follows a specific sequence based on energy levels:
- 1s
- 2s
- 2p
- 3s
- 3p
- 4s
- 3d
- 4p
- 5s
- 4d
- 5p
- 6s
- 4f
- 5d
- 6p
- 7s
- 5f
- 6d
- 7p
Electronic Configuration of Scandium (Sc)
Scandium, with the atomic number 21, has 21 electrons. To determine its electronic configuration, we follow the Aufbau principle and fill the orbitals in order of increasing energy. The electronic configuration of scandium is:
1s2 2s2 2p6 3s2 3p6 4s2 3d1
Let’s break down this configuration:
- 1s2: The first two electrons fill the 1s orbital.
- 2s2: The next two electrons fill the 2s orbital.
- 2p6: The next six electrons fill the 2p orbital.
- 3s2: The next two electrons fill the 3s orbital.
- 3p6: The next six electrons fill the 3p orbital.
- 4s2: The next two electrons fill the 4s orbital.
- 3d1: The final electron fills the 3d orbital.
Importance of Electronic Configuration in Chemistry
The electronic configuration of an element plays a pivotal role in determining its chemical behavior. Here are some key points:
- Chemical Reactivity: The number of valence electrons (electrons in the outermost shell) determines how readily an element will react with others. For example, elements with one or two valence electrons tend to be highly reactive.
- Bonding: The electronic configuration influences the type of bonds an element can form. Elements with partially filled d-orbitals, like scandium, can form complex compounds and exhibit variable oxidation states.
- Physical Properties: The electronic configuration affects properties such as melting point, boiling point, and conductivity. For instance, metals with partially filled d-orbitals often have high melting points and good conductivity.
Electronic Configuration and the Periodic Table
The periodic table is organized based on the electronic configuration of elements. Elements in the same group (column) have similar electronic configurations in their outermost shells, leading to similar chemical properties. For example, all alkali metals (Group 1) have one electron in their outermost s-orbital, making them highly reactive.
Scandium is a transition metal and is located in Group 3 of the periodic table. Transition metals have partially filled d-orbitals, which contribute to their unique chemical and physical properties. The electronic configuration of scandium, with one electron in the 3d orbital, places it in this category.
Electronic Configuration and Oxidation States
The electronic configuration of an element also determines its possible oxidation states. Oxidation states are the hypothetical charges that an atom would have if all bonds were ionic. For scandium, the electronic configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d1 indicates that it can lose up to three electrons to achieve a stable noble gas configuration. Therefore, scandium commonly exhibits oxidation states of +3.
Electronic Configuration and Spectroscopy
Spectroscopy is the study of the interaction between matter and electromagnetic radiation. The electronic configuration of an element influences its spectroscopic properties. For example, the absorption and emission of light by an atom depend on the energy levels of its electrons. Scandium, with its unique electronic configuration, exhibits characteristic spectroscopic signatures that can be used to identify and study its compounds.
Electronic Configuration and Magnetic Properties
The electronic configuration of an element also affects its magnetic properties. Elements with unpaired electrons in their orbitals can exhibit paramagnetism, where they are attracted to magnetic fields. Scandium, with one unpaired electron in the 3d orbital, is paramagnetic. This property is crucial in applications such as magnetic resonance imaging (MRI) and magnetic data storage.
Electronic Configuration and Catalysis
Catalysis is the process by which a substance (catalyst) increases the rate of a chemical reaction without being consumed. The electronic configuration of a catalyst plays a significant role in its effectiveness. Transition metals, including scandium, are often used as catalysts due to their ability to form multiple bonds and stabilize intermediates. The partially filled d-orbitals in scandium allow it to participate in various catalytic reactions, making it valuable in industrial processes.
Electronic Configuration and Alloys
Alloys are mixtures of metals or a metal combined with one or more other elements. The electronic configuration of the constituent elements influences the properties of the alloy. Scandium, when alloyed with other metals like aluminum, can enhance strength, corrosion resistance, and other mechanical properties. The unique electronic configuration of scandium contributes to these improvements, making it a valuable addition to various alloys.
Electronic Configuration and Biological Systems
While scandium is not essential for biological systems, its electronic configuration can provide insights into how metals interact with biological molecules. The partially filled d-orbitals in scandium can form complexes with biological ligands, such as proteins and nucleic acids. Understanding these interactions can help in developing new drugs and therapeutic agents.
Electronic Configuration and Environmental Impact
The electronic configuration of an element also influences its environmental behavior. Scandium, with its unique electronic configuration, can form stable complexes with various environmental ligands. This property can affect its mobility and bioavailability in the environment. Understanding the electronic configuration of scandium can help in assessing its environmental impact and developing strategies for its safe disposal and recycling.
📝 Note: The electronic configuration of an element is a fundamental concept in chemistry that underpins many of its properties and behaviors. Understanding this configuration is crucial for predicting chemical reactivity, bonding, and physical properties.
In summary, the electronic configuration of scandium, with its unique distribution of electrons in the orbitals, plays a crucial role in determining its chemical and physical properties. From its reactivity and bonding behavior to its spectroscopic and magnetic properties, the electronic configuration of scandium influences various aspects of its chemistry. Understanding this configuration is essential for applications in catalysis, alloys, biological systems, and environmental science. By studying the electronic configuration of scandium, we gain insights into the broader principles of chemistry and the behavior of matter at the atomic level.
Related Terms:
- electron configuration for neutral scandium
- electron configuration chart pdf
- electron configuration chart
- full electron configuration for scandium
- sc 3 electron configuration
- scandium electron configuration diagram