Understanding the molecular structure of compounds is fundamental in chemistry, as it helps predict their properties and behaviors. One such compound that has garnered significant interest is boron trifluoride (BF3). This article delves into the Brf3 Molecular Shape, its properties, and its applications, providing a comprehensive overview for both students and enthusiasts.
What is Boron Trifluoride (BF3)?
Boron trifluoride is a chemical compound with the formula BF3. It is a colorless, toxic gas with a pungent odor. BF3 is widely used in various industrial processes, including as a catalyst in organic synthesis and in the production of high-octane gasoline. Its unique properties make it a valuable compound in both academic research and industrial applications.
The Molecular Structure of BF3
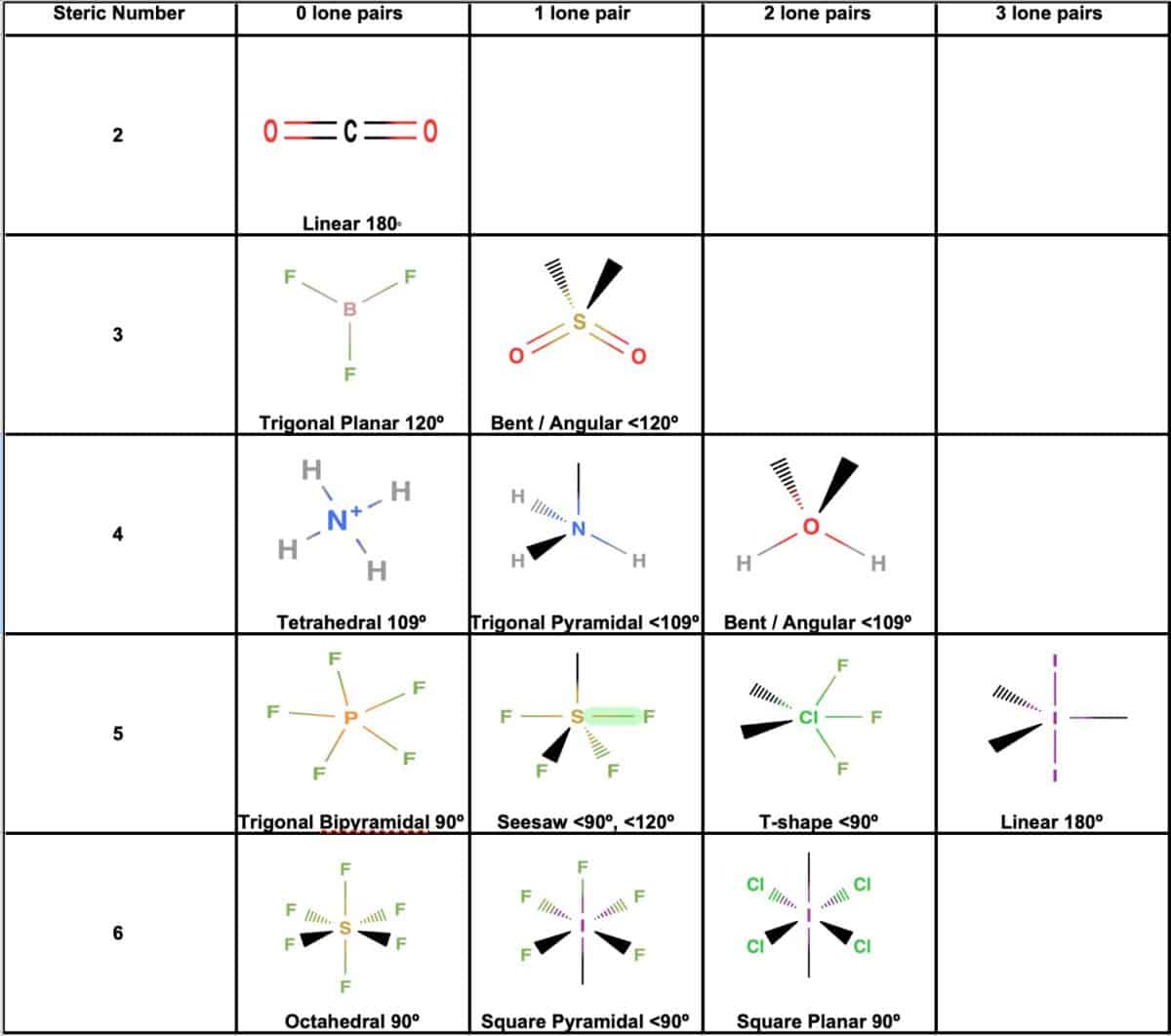
The Brf3 Molecular Shape is a classic example of a trigonal planar geometry. This shape is determined by the Valence Shell Electron Pair Repulsion (VSEPR) theory, which predicts the arrangement of electron pairs around a central atom to minimize repulsion. In BF3, the boron atom is the central atom, and it is bonded to three fluorine atoms.
To understand the Brf3 Molecular Shape, let's break down the bonding and electron arrangement:
- The boron atom has three valence electrons.
- Each fluorine atom contributes one electron to form a covalent bond with boron.
- This results in three covalent bonds around the boron atom, with no lone pairs of electrons.
According to VSEPR theory, the three bonding pairs of electrons will repel each other and arrange themselves in a trigonal planar geometry, with bond angles of 120 degrees. This results in a flat, triangular shape for the BF3 molecule.
Properties of BF3
BF3 exhibits several unique properties that make it useful in various applications. Some of its key properties include:
- Electron Deficiency: BF3 is an electron-deficient compound, meaning it has fewer electrons than needed to form stable bonds. This makes it a strong Lewis acid, readily accepting electron pairs from Lewis bases.
- Toxicity: BF3 is highly toxic and can cause severe health issues if inhaled or absorbed through the skin. Proper safety measures must be taken when handling this compound.
- Reactivity: Due to its electron deficiency, BF3 is highly reactive and can form complexes with various Lewis bases, such as amines and ethers.
- Solubility: BF3 is soluble in organic solvents but reacts with water to form boric acid and hydrofluoric acid.
Applications of BF3
The unique properties of BF3 make it valuable in several industrial and research applications. Some of its key uses include:
- Catalyst in Organic Synthesis: BF3 is widely used as a catalyst in organic synthesis reactions, such as Friedel-Crafts alkylation and acylation.
- Production of High-Octane Gasoline: In the petroleum industry, BF3 is used as a catalyst in the alkylation process to produce high-octane gasoline.
- Laboratory Reagent: BF3 is used in laboratories for various chemical reactions, including the preparation of boron-containing compounds.
- Electronics Industry: BF3 is used in the production of semiconductors and other electronic components due to its ability to form stable complexes with various materials.
Safety Precautions When Handling BF3
Due to its toxicity and reactivity, handling BF3 requires strict safety precautions. Some important safety measures include:
- Use appropriate personal protective equipment (PPE), including gloves, safety glasses, and lab coats.
- Handle BF3 in a well-ventilated area, preferably under a fume hood.
- Avoid contact with water and other reactive substances.
- Store BF3 in a cool, dry place away from incompatible materials.
- In case of accidental exposure, seek immediate medical attention.
🛑 Note: Always follow local regulations and safety guidelines when handling hazardous chemicals like BF3.
Comparing BF3 with Other Boron Compounds
To better understand the Brf3 Molecular Shape and its properties, it’s helpful to compare BF3 with other boron compounds. Here’s a comparison table:
| Compound | Molecular Shape | Electron Deficiency | Common Uses |
|---|---|---|---|
| BF3 | Trigonal Planar | Yes | Catalyst in organic synthesis, production of high-octane gasoline |
| BCl3 | Trigonal Planar | Yes | Catalyst in organic synthesis, production of semiconductors |
| B2H6 | Bridge Structure | Yes | Reducing agent in organic synthesis, production of boron-containing materials |
| B(OH)3 | Trigonal Planar | No | Buffer solutions, flame retardants, glass production |
This comparison highlights the unique properties of BF3 and its Brf3 Molecular Shape, which contribute to its wide range of applications.
Conclusion
Boron trifluoride (BF3) is a fascinating compound with a trigonal planar Brf3 Molecular Shape. Its electron deficiency and reactivity make it a valuable catalyst in various industrial processes. Understanding the molecular structure and properties of BF3 is crucial for its safe and effective use in both research and industrial settings. By following proper safety precautions and handling procedures, the unique properties of BF3 can be harnessed for a wide range of applications, from organic synthesis to the production of high-octane gasoline.
Related Terms:
- brf3 vsepr shape
- brf3 dipole moment
- brf3 vsepr structure
- brf3 polar
- brf3 electron geometry and molecular
- brf3 geometry diagram