Understanding the molecular geometry of compounds is fundamental in chemistry, as it helps predict their properties and behaviors. One such compound that has garnered significant interest is boron trifluoride (BF3). This article delves into the Brf3 Molecular Geometry, exploring its structure, bonding, and the factors that influence its unique properties.
Introduction to Boron Trifluoride
Boron trifluoride (BF3) is a chemical compound consisting of one boron atom bonded to three fluorine atoms. It is a colorless, toxic gas at room temperature and is widely used in various industrial applications, including as a catalyst in organic synthesis and in the production of semiconductors.
Understanding Molecular Geometry
Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule. This arrangement is crucial as it determines the molecule’s reactivity, polarity, and other physical properties. The geometry of a molecule is influenced by the number of electron pairs around the central atom and the repulsion between these pairs.
Brf3 Molecular Geometry: Structure and Bonding
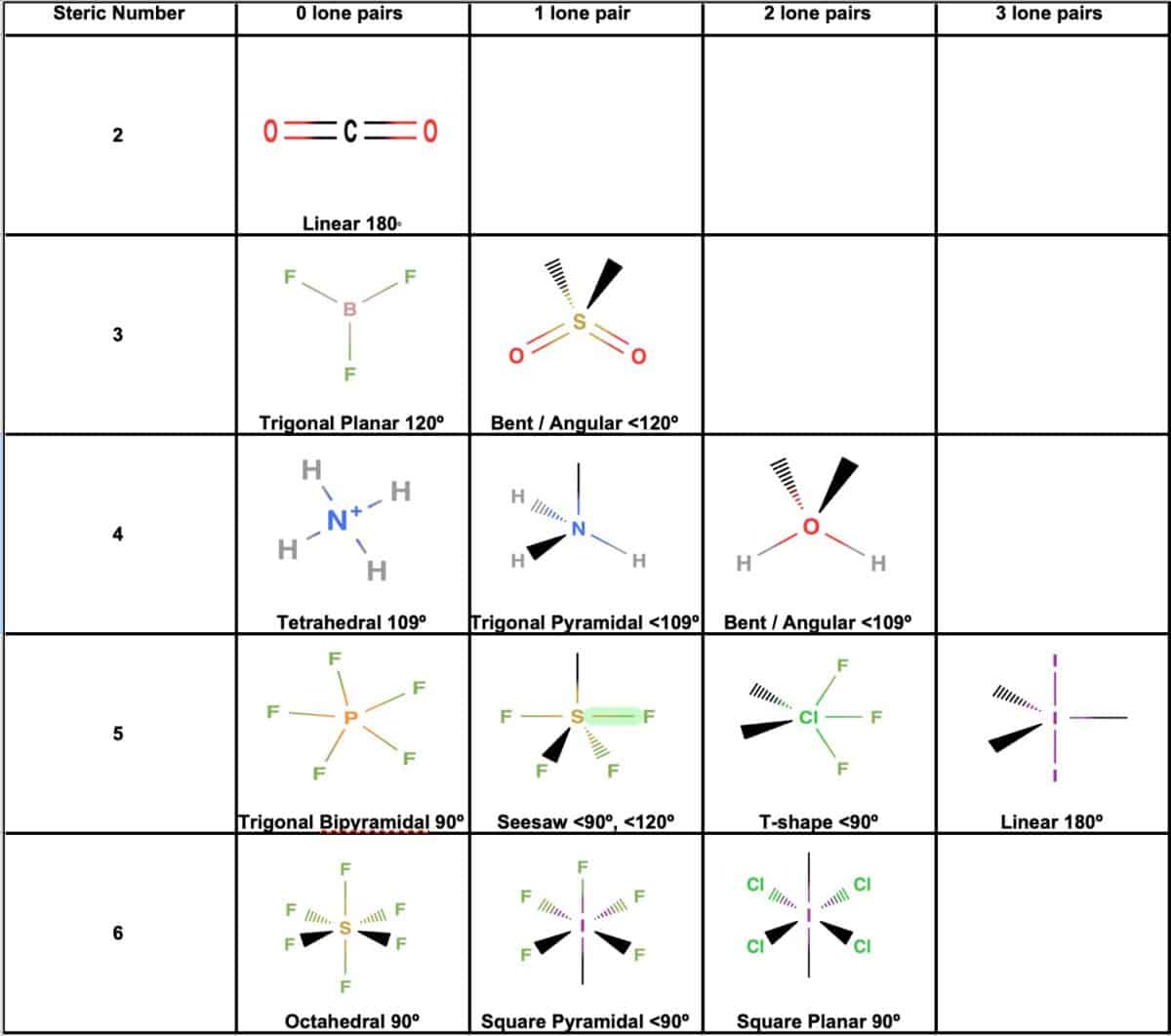
The Brf3 Molecular Geometry is best described as trigonal planar. This means that the boron atom is at the center, with the three fluorine atoms arranged in a plane around it, forming a triangular shape. The bond angles between the fluorine atoms are approximately 120 degrees.
To understand why BF3 adopts this geometry, we need to look at its electronic structure. Boron has three valence electrons, and each fluorine atom contributes one electron to form a covalent bond with boron. This results in three single bonds between boron and each fluorine atom. Since there are no lone pairs of electrons on the boron atom, the repulsion between the bonding pairs is minimized, leading to a trigonal planar geometry.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
The Valence Shell Electron Pair Repulsion (VSEPR) theory is a powerful tool for predicting the geometry of molecules. According to VSEPR theory, electron pairs around a central atom repel each other and arrange themselves in a way that minimizes this repulsion. In the case of BF3, there are three bonding pairs and no lone pairs, leading to a trigonal planar arrangement.
Here is a summary of the VSEPR theory predictions for BF3:
| Central Atom | Number of Bonding Pairs | Number of Lone Pairs | Predicted Geometry |
|---|---|---|---|
| Boron (B) | 3 | 0 | Trigonal Planar |
Factors Influencing Brf3 Molecular Geometry
Several factors influence the molecular geometry of BF3, including:
- Electronegativity: Fluorine is highly electronegative, which means it strongly attracts the shared electrons in the covalent bonds. This polarizes the bonds, making the fluorine atoms partially negative and the boron atom partially positive.
- Size of Atoms: The small size of the fluorine atoms allows them to be closely packed around the boron atom, contributing to the trigonal planar geometry.
- Hybridization: The boron atom in BF3 is sp2 hybridized, which means it uses one s orbital and two p orbitals to form three sp2 hybrid orbitals. These hybrid orbitals are arranged in a trigonal planar geometry.
Properties of Boron Trifluoride
The unique Brf3 Molecular Geometry contributes to several of its properties:
- Polarity: Despite the polar nature of the B-F bonds, the overall molecule is non-polar due to its symmetrical trigonal planar geometry. The dipole moments of the individual bonds cancel each other out.
- Reactivity: BF3 is a strong Lewis acid, meaning it readily accepts electron pairs. This is due to the empty p orbital on the boron atom, which can accommodate additional electrons.
- Stability: The trigonal planar geometry provides stability to the molecule by minimizing electron pair repulsion.
Applications of Boron Trifluoride
Due to its unique properties, BF3 has several important applications:
- Catalyst in Organic Synthesis: BF3 is used as a catalyst in various organic reactions, such as Friedel-Crafts alkylation and acylation.
- Semiconductor Industry: It is used in the production of semiconductors, particularly in the doping process to enhance conductivity.
- Refrigerants: BF3 is used in some refrigeration systems due to its ability to absorb heat efficiently.
🔍 Note: Handling BF3 requires caution due to its toxicity and corrosive nature. Proper safety measures, including the use of personal protective equipment, should be followed when working with this compound.
In conclusion, the Brf3 Molecular Geometry is a fascinating example of how molecular structure influences chemical properties. The trigonal planar geometry of BF3 is a result of its electronic structure and the principles of VSEPR theory. Understanding this geometry is crucial for predicting the behavior of BF3 in various chemical reactions and applications. The unique properties of BF3, such as its Lewis acidity and non-polarity, make it a valuable compound in industrial and chemical processes. By studying the Brf3 Molecular Geometry, we gain insights into the broader principles of molecular structure and reactivity, which are fundamental to the field of chemistry.
Related Terms:
- brf3 polarity
- brf3 electron geometry
- is brf3 polar
- clo3 molecular geometry
- brf5 molecular geometry
- pcl3 molecular geometry